Peer Reviewed
Is It Alzheimer Disease? Guidelines for Evaluating Patients With Mild Memory Loss
AUTHOR:
Anamaria Shanley, MSN, ARNP-BC
CITATION:
Shanley A. Is it Alzheimer disease? Guidelines for evaluating patients with mild memory loss. Consultant. 2016;56(12):1074-1078.
ABSTRACT: Among the many factors that can affect memory among the elderly, the most common is Alzheimer disease (AD). Nevertheless, most patients older than 70 with dementia have never had a clinical cognitive evaluation. Early identification of subtle cognitive changes by way of a neurologic examination and, if needed, specialist referral, may allow for interventions to slow the progression of symptoms and keep an individual in the workforce and independent. Delays in cognitive evaluation can adversely influence comorbidities and delay the diagnosis of AD, which can limit the opportunity for disease stabilization and increase the risk of morbidity. Although treatments to reverse AD are not available, symptomatic treatment options are available, including acetylcholinesterase inhibitors, N-methyl-d-aspartate receptor antagonists, and lifestyle modifications. Efforts to develop better diagnostic tools and disease-modifying therapies for AD are under way.
KEYWORDS: Mild cognitive impairment, memory loss, dementia, Alzheimer disease, acetylcholinesterase inhibitors, N-methyl-d-aspartate receptor antagonists
Primary care providers are on the front lines for detecting the subtle symptoms of cognitive dysfunction. One recent study suggested that more than half of persons older than 70 years with dementia in the United States have never had a clinical cognitive evaluation.1 The authors of another study enrolling 729 physicians in Southern California concluded that the physicians were unaware that more than 40% of their patients had cognitive impairment.2
The possible reasons for why so many patients with mild cognitive dysfunction are not clinically evaluated include the fact that they may not raise concerns about memory loss because they fear losing their independence, or simply that these patients forget that they forget. A spouse or family member, therefore, can be an important resource for supplementing the patient’s history. Moreover, time constraints allow less time for primary care providers to address this topic with patients who present for concerns unrelated to cognition—a dedicated visit likely would be necessary to properly evaluate a patient with concern for memory loss.
Symptoms of cognitive dysfunction can include mild short-term memory loss, the repetition of stories or questions, difficulty with word finding, and new-onset depression or anxiety. Other subtle symptoms are nonadherence to medication regimens, weight loss, and missing or being late to appointments.
Assessment for cognitive impairment is a required element of the annual wellness visit that is covered by Medicare. The use of screening tools and questionnaires can help with this assessment and with time management. The National Institutes of Health’s National Institute on Aging has recommended a number of useful screening tools,3 such as the Ascertain Dementia 8-Item Informant Questionnaire (AD8) and the Mini-Cog, either of which takes 10 minutes or less to complete.
In the elderly population, many factors can affect memory, such as polypharmacy, anxiety, depression, metabolic or endocrine disturbances, insomnia, and more. Once suspicion exists for a dementia diagnosis, it is important to communicate this to the patient and his or her family. Nevertheless, only 45% of older persons or their caregivers are aware of their diagnosis of Alzheimer disease (AD) dementia, compared with 93% of those with a diagnosis of 1 of the 4 most common cancers and 83% of those with a diagnosis of hypertension.4
The following examples represent 2 patients with mild forgetfulness, their evaluations, and their subsequent clinical courses.
Case 1
A 56-year-old woman was referred for a concern for an approximately 3-year history of forgetfulness and difficulty with concentration. The onset was subtle, with a gradual progression. Her symptoms had begun to negatively impact her work performance, and she could no longer perform her job duties effectively.
History. Factors impacting the assessment included a previous diagnosis of fibromyalgia, a history of insomnia, and a diagnosis of obstructive sleep apnea. She had been experiencing mild anxiety, which was contributing to her forgetfulness. She also had a history of migraines, chronic low-back pain, and dysfunctional uterine bleeding that had been treated with oral contraceptives. She had sustained a concussion in a motor vehicle accident 30 years prior. She had no personal history of cardiovascular disease, diabetes, thyroid disease, hypercholesterolemia, cancer, or psychiatric illness. She had no history of tobacco or alcohol use. She had a master’s degree in education.
The patient’s mother had had symptoms of AD dementia and had died in her 60s. Her maternal grandmother had received a clinical diagnosis of vascular dementia. The patient had one sibling, who had an intact memory.
Her current medications included cyclobenzaprine, tramadol, amitriptyline (for insomnia), hormone replacement therapy, baclofen, omeprazole, bupropion, and alprazolam as needed.
Examination. Physical examination findings were unremarkable, with no indication of a movement disorder, apraxia, aphasia, or ataxia. Initial neurologic examination using the Mini-Mental State Examination (MMSE) revealed a score of 22 of 30, suggesting the presence of mild dementia.
Diagnostic tests. Her initial diagnostic workup included magnetic resonance imaging (MRI) of the brain to assess for vascular or structural changes, and blood work (complete blood cell count; comprehensive metabolic panel; thyroid function tests; coagulation tests; assessment of levels of vitamin B12, folate, vitamin D, and homocysteine; neurosyphilis testing; and antinuclear antibody with erythrocyte sedimentation rate testing). The findings of the initial diagnostic workup were unrevealing for any reversible causes to explain the cognitive symptoms. Based on her young age, cerebrospinal fluid (CSF) analysis was completed to rule out acute or chronic infections and an inflammatory or neoplastic process. All test results were normal.
Diagnosis. Given that the patient had normal neurologic workup findings and a first-degree relative with probable AD, an early neurodegenerative process was considered. At her relatively young age (under 65 years), this patient is considered as having early-onset AD.
Discussion. AD is the most common type of neurodegenerative dementia and accounts for 60% to 80% of dementia cases.5 Early-onset AD comprises 5% of the total number of AD cases.6
Alois Alzheimer was a German psychiatrist who developed a passion for neuropathology. He evaluated and treated a 51-year-old woman with symptoms of progressive short-term memory loss and behavioral changes. It is said that Alzheimer became obsessed with her case. When she died 5 years later, Alzheimer performed an autopsy and discovered in her brain the presence of neurofibrillary tau-protein tangles and amyloid plaques. He named the condition “presenile dementia” due to her young age. The condition later would bear Alois Alzheimer’s name.7,8
The National Institute on Aging/Alzheimer’s Association diagnostic guidelines9 identify 3 stages of AD: the asymptomatic preclinical phase, the mild cognitive impairment (MCI) stage, and the dementia stage (Table 1). Based on these guidelines, this patient meets the criteria for MCI due to AD. MCI is considered when mild changes in memory and thinking abilities are noticeable to the person and family members and friends but do not affect the person’s ability to carry out everyday activities.10,11

While the gold standard for definitive diagnosis of AD is by way of a brain biopsy, the clinical diagnosis of AD using simple screening measures such as the MMSE has a sensitivity ranging between 39% and 98%.12 The diagnostic reliability of the CSF AD biomarkers (reduced levels of amyloid-β 1-42 and increased levels of total tau and phosphorylated tau) is still limited and therefore not yet recommended for routine use.13 Between 2012 and 2014, the US Food and Drug Administration (FDA) approved 3 amyloid-β labeling cerebral positron emission tomography (PET) scans for clinical use in the evaluation of individuals with cognitive impairment. Sensitivity of the scans ranges from 96% to 97%, with specificity of 100%. The PET scans currently are not covered by Medicare.14
There is no cure for AD, nor are there any FDA-approved treatments for individuals with MCI related to AD. Two medication classes have been FDA-approved for the treatment of AD, acetylcholinesterase inhibitors and N-methyl-d-aspartate (NMDA) receptor antagonists (Table 2).
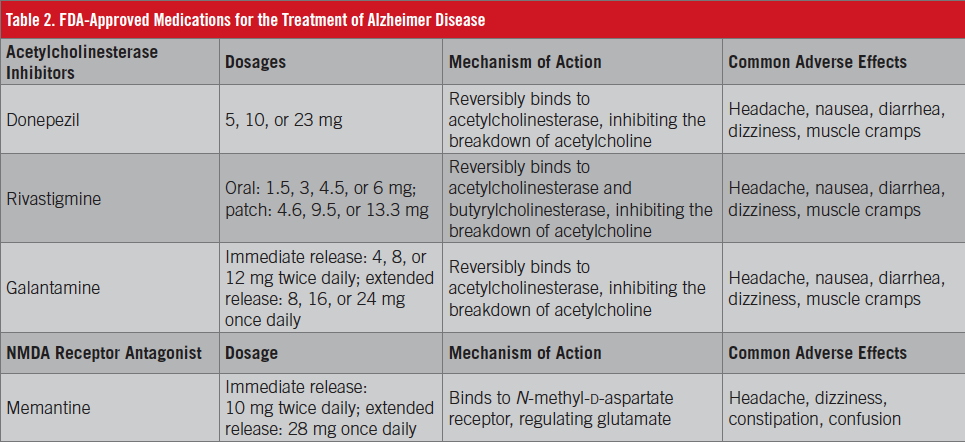
The acetylcholinesterase inhibitors are approved for mild to moderate and moderate to severe AD. The 3 available drugs in this class are donepezil, rivastigmine (oral and patch), and galantamine. These medications block acetylcholinesterase, inhibiting the breakdown of acetylcholine, which is essential for cognitive processing and memory. In AD, the acetylcholine-producing neurons (cholinergic neurons) are affected, therefore reducing acetylcholine production.15 The efficacy of the 3 agents is approximately the same based on the results of head-to-head trials.15,16 Among the most common reported adverse effects of the medication class are headache, nausea, dizziness, muscle cramps, and diarrhea likely related to increased cholinergic activity.17-19 There is no evidence acetylcholinesterase inhibitors have any disease-modifying effects. It is important to discuss realistic expectations of these medications, since they do not change the disease progression.
Memantine, an NMDA receptor antagonist, also is FDA-approved for the treatment of mild to severe AD. The mechanism of action of memantine differs from that of acetylcholinesterase inhibitors in that memantine modulates the NMDA receptor (regulating glutamate), which is felt to be overexcited in AD.20 Memantine is generally well tolerated, with relatively few adverse effects, which include hypertension, dizziness, headache, confusion, and constipation.21 Memantine can be used as monotherapy as well as in combination with a acetylcholinesterase inhibitor.
Treatment and follow-up. Over the course of continued follow-up care, our patient’s symptoms continued to progress. The decision was made to initiate memantine therapy.
Case 2
A 66-year-old man was referred with a 4-year history of progressive forgetfulness. During a previous evaluation at a large multispecialty institution, his symptoms had been attributed to depression.
A neuropsychological evaluation identified cognitive impairment meeting the criteria for MCI due to AD. He was started on symptomatic therapy with a rivastigmine patch, which later was switched to donepezil out of concern for apparent treatment failure with rivastigmine. At a later follow-up visit, memantine was added to the man’s pharmacotherapy regimen
After his initial consultation, which included a review of his previous neurologic workup results (brain MRI, blood work, and neuropsychological testing), the patient was offered enrollment in an 18-month, double-blind, placebo-controlled clinical trial of a disease-modifying anti-amyloid-β monoclonal antibody.
Throughout the patient’s follow-up visits over the course of the next year, his office memory test results remained stable. He continues on a biannual schedule of follow-up visits and since has been enrolled in another clinical trial for a disease-modifying drug.
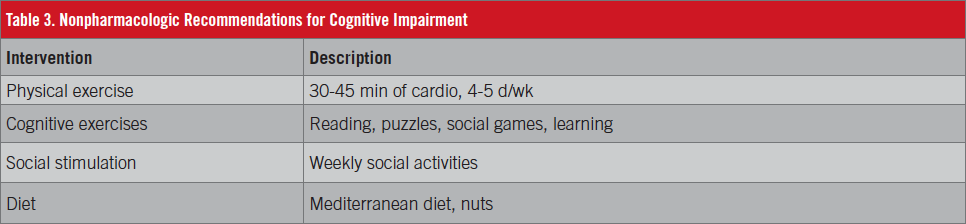
Discussion. Lifestyle behaviors should also be discussed with patients who have cognitive impairment. Studies support that regular exercise, a healthy diet, and cognitive activities benefit cognition.
Rebok and colleagues22 studied the effects of cognitive training on individuals aged 65 and older who had little memory impairment with no effect on function. The treatment groups received structured cognitive training for 5 to 6 weeks, followed by booster training at 11 and 35 months. During retesting 10 years later, each intervention group reported less difficulty with independent activities of daily living. At the 10-year follow up, patients showed a persistent benefit in the areas of reasoning and speed. Another study using postmortem brain pathology to assess the effects of cognitive stimulation throughout the lifespan found evidence that individuals who were more cognitively active in middle age and late life had a slower rate of cognitive decline, independent of neuropathology.23
The results of the randomized, controlled FINGER trial (Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability)24 showed that lifestyle intervention was associated with significant improvement in cognition. Study participants were assigned to various intervention groups: dietary counseling (increasing intake of fruits, vegetables, whole grains, lean protein, and healthy fats), aerobic exercise and weight training, and cognitive training with computer programs several times per week. Compared with placebo, the intervention groups experienced a 25% improvement in overall Cardiovascular Risk Factors, Aging, and Incidence of Dementia (CAIDE) scores.
A meta-analysis by Xu and colleagues25 identified several risk factors for developing AD, including hyperhomocysteinemia, depression, preexisting disease (frailty, carotid atherosclerosis, hypertension, low diastolic blood pressure, and type 2 diabetes), low education, and high body mass index in midlife. Protective factors identified included the use of certain medications (estrogen, statins, antihypertensives, and nonsteroidal anti-inflammatory drugs), certain dietary exposures (folate, vitamin E, vitamin C, and coffee), and cognitive activity.
Multiple studies have tested the relationship between diet and cognitive health, including a study investigating the effects of a Mediterranean diet with olive oil and nuts that demonstrated benefits in cognitive testing results.26
Early Identification of AD
Early identification of subtle cognitive change is important. A delayed cognitive evaluation can affect comorbidities, delay diagnosis, and limit the opportunity for stabilization, possibly negatively impacting a person’s personal safety.27 Even though disease-modifying medications for AD are not yet available, options for symptomatic treatment include nonpharmacologic lifestyle modifications (Table 3), and enrollment in clinical trials if appropriate. Early intervention may delay the progression of symptoms, keeping the individual in the workforce or independent and ultimately reducing costs and optimizing quality of life.
In 2011, the president signed the National Alzheimer’s Project Act into law. The law requires the National Alzheimer’s Project to do the following28:
- Create and maintain an integrated national plan to address AD
- Coordinate AD research and services across all federal agencies
- Accelerate the development of treatments that would prevent, halt, or reverse the course of AD
- Improve early diagnosis and coordination of care and treatment of AD
- Decrease disparities in AD for ethnic and racial minority populations at higher risk for AD
- Coordinate with international bodies to fight AD globally
The national plan to address AD has set a goal to provide effective diagnostic testing and treatment for individuals with symptoms of AD by 2025.28 All US clinical drug trials must be registered, and can be found at either https://www.clinicaltrials.gov/ or https://www.nia.nih.gov/alzheimers/clinical-trials. Patients who are suspected of having symptoms of AD are encouraged to explore enrollment in a clinical trial if it is appropriate for them.
The annual Alzheimer’s Association International Conference reports the status of the numerous clinical trials of medications for the potential treatment of AD. There have been several reports by the sponsors that indicate that the compounds targeting β-amyloid plaques may provide cognitive stability.29
The National Institute on Aging, in partnership with several philanthropic organizations and a pharmaceutical company, seeks to enroll 1000 asymptomatic individuals in a secondary prevention trial called the A4 study (Anti-Amyloid Treatment in Asymptomatic Alzheimer’s).30 The study seeks ways to prevent or delay symptoms of dementia in individuals who are considered at high risk for developing AD as identified by elevated brain amyloid levels on amyloid-β labeling cerebral PET scans.
The American College of Radiology Imaging Network and the Alzheimer’s Association are sponsoring the IDEAS (Imaging Dementia–Evidence for Amyloid Scanning) study. For this diagnostic trial, these expert groups are working with the Centers for Medicare and Medicaid Services to enroll more than 18,000 Medicare beneficiaries over 24 months with symptoms of memory loss or dementia. The purpose of the study is to assess the clinical usefulness of amyloid imaging in the diagnosis of AD in patients with MCI.31
Anamaria Shanley, MSN, ARNP-BC, is a nurse practitioner at the Compass Clinic in Orlando, Florida.
References:
- Kotagal V, Langa KM, Plassman BL, et al. Factors associated with cognitive evaluations in the United States. Neurology. 2015;84(1):64-71.
- Chodosh J, Petitti DB, Elliott M, et al. Physician recognition of cognitive impairment: evaluating the need for improvement. J Am Geriatr Soc. 2004;52(7):1051-1059.
- Instruments to detect cognitive impairment in older adults. National Institute on Aging. https://www.nia.nih.gov/research/cognitive-instrument. Accessed November 3, 2016.
- Factsheet: 2015 Alzheimer’s disease facts and figures. Alzheimer’s Association. https://www.alz.org/facts/downloads/ff_quickfacts_2015.pdf. Published March 2015. Accessed November 3, 2016.
- Alzheimer’s & dementia, types of dementia. Alzheimer’s Association. http://www.alz.org/dementia/types-of-dementia.asp. Accessed November 3, 2016.
- Younger/early onset Alzheimer’s & dementia. Alzheimer’s Association. http://www.alz.org/alzheimers_disease_early_onset.asp. Accessed November 3, 2016.
- Alois Alzheimer. Biography.com. http://www.biography.com/people/alois-alzheimer-21216461. Updated February 25, 2016. Accessed November 3, 2016.
- Tagarelli A, Piro A, Tagarelli G, Lagonia P, Quattrone A. Alois Alzheimer: a hundred years after the discovery of the eponymous disorder. Int J Biomed Sci. 2006;2(2):196-204.
- Jack CR Jr, Albert MS, Knopman DS, et al. Introduction to the recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):257-262.
- New diagnostic criteria and guidelines for Alzheimer’s disease. Alzheimer’s Association. http://www.alz.org/research/diagnostic_criteria/. Accessed November 3, 2016.
- Albert MS, DeKosky ST, Dickson D, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):270-279.
- Wollman DE, Prohovnik I. Sensitivity and specificity of neuroimaging for the diagnosis of Alzheimer’s disease. Dialogues Clin Neurosci. 2003;5(1):89-99.
- Mattsson N, Andreasson U, Persson S, et al; Alzheimer’s Association QC Program Work Group. CSF biomarker variability in the Alzheimer’s Association quality control program. Alzheimers Dement. 2013;9(3):251-261.
- Decision memo for beta amyloid positron emission tomography in dementia and neurodegenerative disease (CAG-00431N). Centers for Medicare and Medicaid Services. https://www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=265. Accessed November 3, 2016.
- Francis PT. The interplay of neurotransmitters in Alzheimer’s disease. CNS Spectr. 2005;10(11 suppl 18):6-9.
- Wilkinson DG, Francis PT, Schwam E, Payne-Parrish J. Cholinesterase inhibitors used in the treatment of Alzheimer’s disease: the relationship between pharmacological effects and clinical efficacy. Drugs Aging. 2004;21(7):453-478.
- Aricept [package insert]. New York, NY: Pfizer Inc; 2015.
- Exelon [package insert]. East Hanover, NJ: Novartis Pharmaceuticals Corp; 2015.
- Razadyne [package insert]. Titusville, NJ: Janssen Pharmaceuticals Inc; 2013.
- Tampi RR, van Dyck CH. Memantine: efficacy and safety in mild-to-server Alzheimer’s disease. Neuropsychiatr Dis Treat. 2007;3(2):245-258.
- Namenda [package insert]. St Louis, MO: Forest Pharmaceuticals Inc; 2014.
- Rebok GW, Ball K, Guey LT, et al; ACTIVE Study Group. Ten-year effects of the Advanced Cognitive Training for Independent and Vital Elderly cognitive training trial on cognition and everyday functioning in older adults. J Am Geriatr Soc. 2014;62(1):16-24.
- Wilson RS, Boyle PA, Yu L, Barnes LL, Schneider JA, Bennett DA. Life-span cognitive activity, neuropathologic burden, and cognitive aging. Neurology. 2013;81(4):314-321.
- Ngandu T, Lehtisalo J, Solomon A, et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet. 2015;385(9984):2255-2263.
- Xu W, Tan L, Wang H-F, et al. Meta-analysis of modifiable risk factors for Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 2015;86(12):1299-1306.
- Valls-Pedret C, Sala-Vila A, Serra-Mir M, et al. Mediterranean diet and age-related cognitive decline: a randomized clinical trial. JAMA Intern Med. 2015;175(7):1094-1103.
- McPherson S, Schoephoester G. Screening for dementia in a primary care practice. Minn Med. 2012;95(1):36-40.
- National Plan to Address Alzheimer’s Disease: 2016 Update. Washington, DC: US Dept of Health and Human Services; 2015. https://aspe.hhs.gov/report/national-plan-address-alzheimers-disease-2016-update. Accessed November 3, 2016.
- Clinical trials results and new data analyses in amyloid-related therapies from the Alzheimer’s Association International Conference 2015 [press release]. Washington, DC: Alzheimer’s Association; July 22, 2015. https://www.alz.org/aaic/_downloads/Wed-7am.pdf. Accessed November 3, 2016.
- What is the A4 study? A4 Study. http://a4study.org/about/. Accessed November 3, 2016.
- IDEAS Study. http://www.ideas-study.org. Accessed November 3, 2016.


