Facial Plaques in a 4-Year-Old Girl
A 4-year-old Latina presented with red, indurated plaques that had been present on her cheeks and dorsal nose for 3 months. The girl’s mother reported that the lesions worsened with sun exposure and had progressed slowly despite twice daily application of a topical antifungal cream.
The child was otherwise healthy, and her routine vaccinations were current. Review of systems was unremarkable aside from easily bruising and bleeding, according to the girl’s mother. The child was not taking any oral medication. There was no history of travel outside of the United States, and other family member were healthy and without rash.

On physical examination, the patient had erythematous, raised plaques without discernible scale on the cheeks and nasal dorsum (Figures 1-3). The remainder of the skin and mucosal examination findings were unremarkable.
What is the cause of these lesions?
(Answer and discussion on next page)
Answer: Lupus erythematosus tumidus
Results of potassium hydroxide prep testing of skin scrapings from the lesion were negative for fungal elements. One 4-mm punch biopsy from the right cheek was obtained and submitted for hematoxylin and eosin staining.
A complete blood count (CBC), complete metabolic panel (CMP), liver function tests (LFTs), coagulation tests (prothrombin time [PT], activated partial thromboplastin time [aPTT], and international normalized ratio [INR]), serum antinuclear antibody (ANA) titer, serum anti-Ro/SSA antibody titer, total hemolytic complement (CH50) assay, complement components C3 and C4 assay, and C-reactive protein (CRP) test were ordered.

Figure 1. Indurated, erythematous plaques had been present for 3 months on a 4-year-old girl’s cheeks and dorsal nose.
Lupus erythematosus tumidus (LET) is a variant of chronic cutaneous lupus that is characterized by indurated, erythematous plaques without discernible scale or follicular plugging. Most commonly, LET affects postpubertal patients between the ages of 20 and 40 years, with men and women equally affected.

Figure 2. The lesions had no discernible scale or follicular plugging.
Clinical Presentation
LET lesions most commonly are observed in areas of sun exposure (eg, the face, the upper back, the V area of the neck, the extensor aspects of the arms and shoulders), while sun-spared areas (eg, the inner aspect of the arms, the axillae), knuckles, and lower extremities are not affected. Once a lesion has developed, it can disappear spontaneously within days to weeks. However, lesions often are reproducible with sun exposure and provocative UV-A (315-400 nm) and UV-B (280-315 nm) radiation phototesting.1
Morphology of the lesions is similar to Jessner lymphocytic infiltration (some believe these entities to be the same or closely related2), and the lesions may develop central clearing similar to that of the annular variant of subacute cutaneous lupus erythematosus (SCLE). Lesions tend to heal without scarring or atrophy, because the dermal-epidermal and follicular basal layers and the subcutaneous fat are not inflamed.
Histopathologic examination results show lymphohistiocytic infiltrate of the dermis with increased mucin.3 Patients presenting with LET rarely have the other cutaneous findings associated with systemic lupus erythematosus (SLE), such as hypopigmentation, mucous membrane ulcers, diffuse alopecia, or livedo reticularis or vasculitides. Patients presenting with LET do not have evidence of systemic involvement1 characterized by 4 or more of the 11 American Rheumatism Association (now the American College of Rheumatology) criteria for the diagnosis of SLE.4
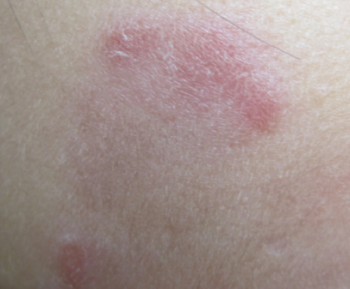
Figure 3. Some of the lesions featured central clearing similar to that of the annular variant of subacute cutaneous lupus erythematosus.
Differential Diagnosis
In addition to LET, the differential diagnosis in the patient described here included discoid lupus erythematosus, SCLE, Jessner lymphocytic infiltration, polymorphic light eruption, pseudolymphoma, tinea faciei, allergic contact dermatitis, and malar eruption of Bloom syndrome.
Treatment
Patients with LET should be screened for underlying SLE with serum ANA and anti-Ro/SSA titers, along with basic laboratory testing if clinically indicated. UV exposure should be avoided with the of protective clothing (eg, broad-brimmed hat, high-collared shirt), the avoidance of prolonged outdoor activity between 11 am and 3 pm, and the use of broad-spectrum sunscreen.
Approximately half of patients with LET will experience clinical resolution with UV avoidance and topical or intralesional corticosteroid therapy, the strength of which is determined by location on the body.1 Patients without clinical resolution with topical therapy alone often improve with the addition of oral antimalarial agents. Hydroxychloroquine 6.0 to 6.5 mg/kg daily has been used successfully.3 Before starting oral antimalarial therapy, patients should undergo basic laboratory testing (ie, CBC, CMP, LFTs) and a complete baseline ophthalmologic examination. While on oral antimalarial therapy, patients should be monitored every 4 to 6 months for rare agranulocytosis and hemolytic anemia. Additionally, patients should be monitored for changes in vision (eg, blurring, halos, central scotomata) and cutaneous hyperpigmentation (blue-gray darkening of the shins, mucosa, and sun-exposed areas).5
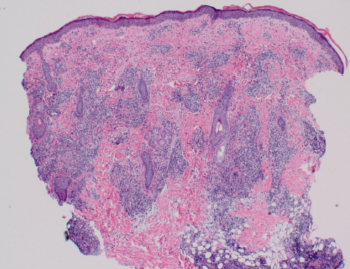
Figure 4. Histopathologic examination results showed superficial and deep lymphocytic infiltration in a perivascular and periadnexal distribution pattern.
Our Patient
Our patient’s clinical presentation was most consistent with LET. Histopathologic examination results showed superficial and deep lymphocytic infiltration in a perivascular and periadnexal distribution (Figure 4). On closer inspection, widening of collagen spacing with a blue, wispy material most consistent with dermal interstitial mucin deposition was seen (Figure 5). Colloidal iron stain confirmed the presence of increased interstitial mucin (Figure 6). There was no follicular plugging, dermoepidermal junction involvement, or epidermal change (eg, hyperkeratosis, acanthosis) (Figure 7).
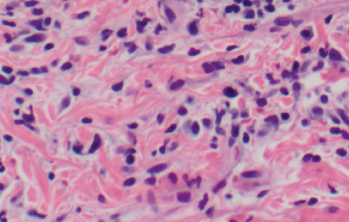
Figure 5. Widened collagen spacing with a blue, wispy material consistent with dermal deposition of interstitial mucin.
Results of the CBC, CMP, LFTs, CRP, and coagulation tests were within normal limits. The serum ANA titer and the anti-Ro/SSA antibody were negative. Results of CH50, C3, and C4 assays were within normal limits.
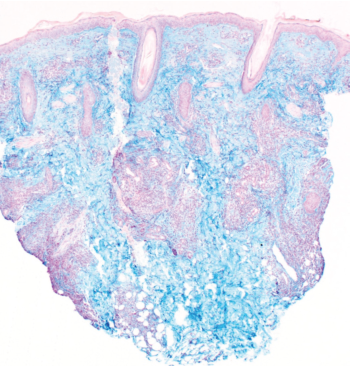
Figure 6. Colloidal iron stain confirmed the presence of increased interstitial mucin.
We chose to order complement studies in this patient despite a low clinical suspicion for systemic disease, because localized lupus-like skin disease is a presenting sign of inherited complement deficiencies in young, prepubescent children. Deficiency of complement C1q, the first component of the classical pathway, and other early classic pathway complement components (C2 and C4) is associated with the development of lupus-like disease, including localized skin disease, nephritis, and central nervous system disease, likely because of ineffective antibody-antigen complexes clearance.6 Without these early complement components, antibody-antigen complexes are not cleared and can deposit in end organs and tissues, causing tissue injury; these antibody-antigen complexes in turn may stimulate autoantibody generation in various organs and tissues.6
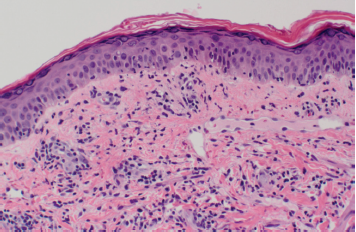
Figure 7. Hematoxylin and eosin staining showed the absence of dermoepidermal junction involvement.
Our patient has experienced clinical improvement with aggressive UV avoidance and topical triamcinolone 0.1% cream twice daily for 2 weeks, with transition to hydrocortisone valerate 0.2% cream twice daily for 2 weeks. Intralesional therapy and/or an oral antimalarial agent will be considered if complete clearance is not achieved with topical therapy alone.
References:
1. Kuhn A, Richter-Hintz D, Oslislo C, Ruzicka T, Megahed M, Lehmann P. Lupus erythematosus tumidus: a neglected subset of cutaneous lupus erythematosus: report of 40 cases. Arch Dermatol. 2000;136(8):1033-1041.
2. O’Toole EA, Powell F, Barnes L. Jessner’s lymphocytic infiltrate and probable discoid lupus erythematosus occurring separately in two sisters. Clin Exp Dermatol. 1999;24(2):90-93.
3. Stead J, Headley C, Ioffreda M, Kovarik C, Werth V. Coexistence of tumid lupus erythematosus with systemic lupus erythematosus and discoid lupus erythematosus: a report of two cases of tumid lupus. J Clin Rheumatol. 2008;14(6):338-341.
4. Tan EM, Cohen AS, Fries JF, et al. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1982;25(11):1271-1277.
5. Samanta A, Goh L, Bawendi A. Are evidence-based guidelines being followed for the monitoring of ocular toxicity of hydroxychloroquine? A nationwide survey of practice amongst consultant rheumatologists and implications for clinical governance. Rheumatology (Oxford). 2004;43(3):346-348.
6. Vassallo G, Newton RW, Chieng SE, Haeney MR, Shabani A, Arkwright PD. Clinical variability and characteristic autoantibody profile in primary C1q complement deficiency. Rheumatology (Oxford). 2007;46(10):1612-1614.
Dr Dougherty is in the Department of Dermatology and Dr Stetson is chair of the Department of Dermatology and director of the residency program at Texas Tech University Health Sciences Center in Lubbock.
Kirk Barber, MD, FRCPC––Series Editor:Dr Barber is a consultant dermatologist at Alberta Children’s Hospital and clinical associate professor of medicine and community health sciences at the University of Calgary in Alberta.
Editor’s note: A version of this article was originally published in The Dermatologist (Dougherty AL, Stetson CL. What are these facial plaques in this 4-year-old child? 2013;21[8]:47-50).


