Devices for Heart Rhythm Disorders in the Elderly
This article is the fifth in a continuing series on cardiovascular issues in the older adult. The fourth article in the series, “The Elderly and Peripheral Arterial Disease,” was published in the July 2011 issue of Clinical Geriatrics. The remaining articles in the series, to be published in future issues of the Journal, will discuss such topics as heart failure and hypertension.
______________________________________________________________________________________________
Heart rhythm disorders, or arrhythmias, are very common in the elderly population. They can be categorized as tachyarrhythmias (an abnormally rapid heart rhythm) or bradyarrhythmias (an abnormally slow heart rhythm), depending on the presenting heart rate. A significant number of elderly patients with such rhythm disorders will require implantation of a cardiac rhythm device. The choice between a pacemaker or an implantable cardioverter-defibrillator (ICD) with or without cardiac resynchronization therapy (CRT) will depend on a number of factors related to the patient’s heart condition, symptoms, and the presenting arrhythmia. Although multiple clinical trials have been conducted in patients with heart failure undergoing different forms of device therapy, older Americans are largely underrepresented in these studies.1 This article reviews the current guidelines on device implantation, with a focus on the use of pacemakers, ICD therapy, and CRT in the elderly population.
Pacemaker Therapy in the Elderly
Aging is often associated with progressive fibrosis of the cardiac conduction system, including the sinoatrial and atrioventricular (AV) nodes. This leads to the development of bradyarrhythmias, which often necessitate pacemaker therapy. More than 80% of all pacemakers are implanted in patients >65 years of age,2 and up to 14% are implanted in patients >85 years of age.3 Knowledge of the appropriate indications for pacemaker implantation in the elderly population and the appropriate management of an elderly individual with a pacemaker is important for healthcare providers treating this population.
Sinus Node Dysfunction
Sinus node dysfunction (SND) most commonly occurs in the seventh and eighth decades of life4-6 and is typically caused by senescence of the sinus node. However, SND may occur at any age as a result of any condition that destroys sinus node cells, such as ischemia or infarction, infiltrative disease, collagen vascular disease, surgical trauma, endocrine abnormalities, and autonomic insufficiency. It may also be caused by some drug therapies, such as beta blockers or other antiarrhythmic agents that may suppress sinus node function. Patients with SND may exhibit asymptomatic sinus bradycardia or may experience symptoms of syncope due to sinus pauses or exertional fatigue due to chronotropic incompetence (defined below).
The indications for permanent pacing due to SND, manifesting as sinus bradycardia or pauses, require the presence of symptoms such as exertional fatigue or syncope. There is no absolute rate of bradycardia or duration of pause that requires pacemaker implantation in the absence of symptoms. In fact, the current American College of Cardiology (ACC)/American Heart Association (AHA)/Heart Rhythm Society (HRS) 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities6 state that permanent pacemaker implantation is not indicated for SND in asymptomatic patients. Therefore, it is imperative that any patient with evidence of SND be questioned about the presence of symptoms attributable to this dysfunction.
While some individuals with SND may present with syncope, patients more commonly present with exertional fatigue. Patients may be asymptomatic with a heart rate of 40 beats per minute at rest, but they may develop significant symptoms of exertional intolerance if their heart rate fails to augment upon exertion. This phenomenon, referred to as chronotropic incompetence, is frequently seen in the elderly population but is often overlooked. Older individuals often disregard symptoms of fatigue due to SND, falsely attributing them to their age. Further, the onset of these symptoms may be insidious, slowly worsening over a period of months to years. It is therefore important to ask patients to describe their exercise tolerance over a period of time, particularly how their current exercise tolerance compares with that of the previous 1 or 2 years.
When assessing our patients for chronotropic incompetence, we often ask them to walk around our clinic or up a flight of steps (if possible) to calculate their heart rate response to exercise. If they develop exercise-limiting fatigue or dyspnea without their heart rate increasing to >100 beats per minute (or higher for younger patients), the diagnosis of chronotropic incompetence must be considered. In cases where a walk in the clinic or up a flight of steps results in equivocal results, a formal exercise test using the standard Bruce Protocol, or the less strenuous Modified Bruce Protocol or Naughton Protocol, may be performed.
In addition to chronotropic incompetence, patients may present with symptoms of intermittent SND, such as syncope. SND often manifests as prolonged sinoatrial pauses, particularly after the termination of atrial tachyarrhythmias, or may manifest as sinus bradycardia, both of which may limit the ability to titrate medications necessary for the control of a paroxysmal tachycardia. This phenomenon of intermittent SND and atrial tachyarrythmias, often referred to as tachy-brady syndrome, can frequently be seen after conversion of atrial fibrillation to sinus rhythm. During an atrial tachyarrhythmia, such as atrial fibrillation or other supraventricular tachycardia, the sinus node activity is suppressed. Upon termination of the arrhythmia, the sinus node requires a period of time to recover. In elderly patients, this sinus node recovery time may be prolonged to such a degree that the patient experiences pauses of sufficient duration to cause symptoms. While there is no absolute length of sinus pause that requires pacemaker placement, patients with pauses of sufficient duration to cause symptoms should be considered for pacemaker implantation.
As noted previously, drug therapy may result in SND. In cases of drug-induced SND, pacemaker implantation may be considered if the withdrawal of the offending agent does not correct the SND or if the drug is medically necessary and cannot be discontinued.
Heart Block
Indications for pacemaker implantation in patients with heart block are dependent upon the likelihood that the AV block will be permanent, the likelihood of progression to complete heart block and asystole, and the presence of symptoms. Patients with temporary heart block, as may occur secondary to electrolyte abnormalities, Lyme disease, or vagal triggers such as temporary hypoxia during sleep apnea, typically do not require pacing. Conversely, heart block due to diseases such as sarcoidosis, amyloidosis, and neuromuscular diseases is often treated with a permanent pacemaker, as it is unlikely to resolve and more frequently progresses to complete heart block.7
AV block may occur at the level of the AV node or at or below the His bundle. It is classified as first-, second-, or third-degree/complete heart block, depending upon the relationship between the P waves and QRS complexes. Third-degree AV block (complete heart block) is defined as absence of AV conduction with dissociation of P waves and QRS complexes; it is usually treated with pacemaker implantation. First-degree AV block is defined as a prolongation of the PR interval to >200 milliseconds. First-degree AV block is usually not associated with symptoms and typically does not necessitate implantation of a permanent pacemaker or initiation of another therapy.6
Second-degree AV block is subclassified as Mobitz type I (also known as Wenckebach block) and Mobitz type II block. Mobitz type I second-degree AV block is characterized by progressive prolongation of the PR interval prior to a nonconducted P wave and a shorter PR interval following the first P wave to resume conduction to the ventricle. It is usually seen in conjunction with a narrow QRS complex. Type I second-degree AV block is due to delayed conduction in the AV node, and progression to complete heart block is uncommon.7,8 While more commonly seen in the elderly population, type I second-degree AV block can be demonstrated in all patients with high atrial rates. Thus, pacemaker implantation is typically not indicated for type I second-degree AV block unless it leads to significant symptoms, such as exertional fatigue.
Mobitz type II second-degree AV block is characterized by fixed PR intervals before and after nonconducted P waves, and is typically seen in conjunction with a wide QRS complex. Mobitz type II second-degree AV block has a much higher likelihood of progressing to complete heart block and therefore is often an indication for pacemaker implantation, even in the absence of symptoms, particularly if it occurs in the setting of a bifascicular or left bundle branch block. An alternating bundle branch block suggests significant disease in the distal conduction system and is therefore typically an indication for permanent pacemaker implantation.6
Patients are frequently referred for pacemaker evaluation because of pauses noted during cardiac monitoring. Pacemaker implantation should be considered in the following patients: individuals with symptomatic pauses due to AV block; awake, symptom-free patients in sinus rhythm who exhibit complete or Mobitz type II second-degree heart block and have documented periods of asystole of ≥3 seconds; and patients who experience pauses of >5 seconds while awake during episodes of atrial fibrillation, even if these pauses are deemed to be asymptomatic, as this is often an indicator of advanced conduction system disease. Occasionally, patients may experience pauses secondary to carotid sinus hypersensitivity. These patients are candidates for permanent pacing if they have recurrent syncope due to carotid sinus stimulation inducing ventricular asystole of >3 seconds.6 There are also data to suggest that elderly patients with recurrent unexplained falls may benefit from permanent pacemaker implantation if they exhibit evidence of carotid sinus hypersensitivity.9
In addition to the aforementioned indications for pacing, the ACC/AHA/HRS guidelines recommend permanent pacemaker implantation for any patient with symptomatic heart block and for the treatment of arrhythmias or other medical conditions that require drug therapy—which itself results in symptomatic bradycardia or heart block.6 Patients who exhibit third-degree or advanced second-degree AV block (Mobitz type II) during exercise should also be considered for pacemaker implantation.
Implantable Cardioverter-Defibrillator and Cardiac Resynchronization Therapy in the Elderly
With current advances in the management of heart failure and other coexisting illnesses, a growing number of older individuals will become candidates for ICD therapy. In addition, a growing population of older patients with heart failure symptoms and low ejection fraction (<30%-35%) despite optimal medical therapy will become suitable candidates to receive CRT as an integral part of the management of their heart failure.
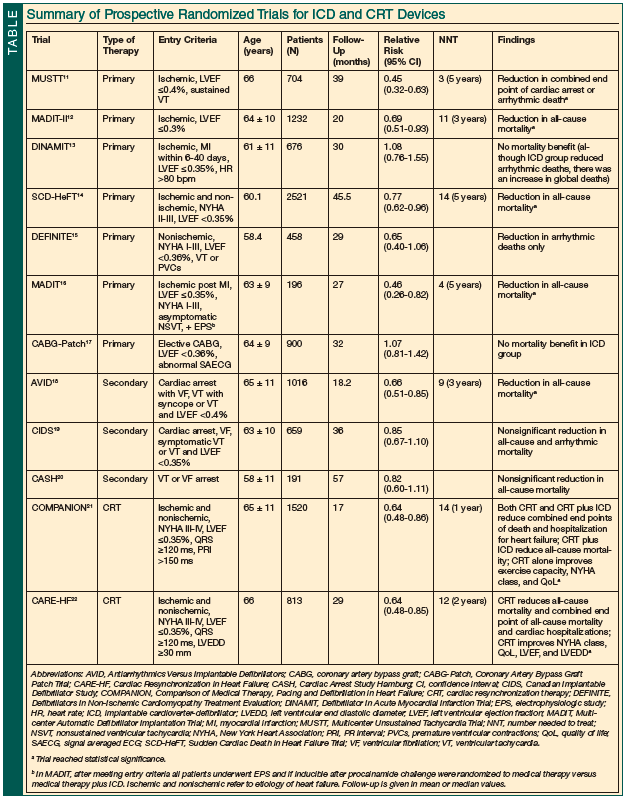
Despite the fact that a large proportion of recipients of ICD and CRT devices in the United States are >70 years of age,10 this patient population was significantly underrepresented in the large, randomized controlled trials assessing cardiac rhythm devices, yet the results of these trials became the basis for primary and secondary prevention ICD implantation indications. (The Table provides a summary of all major primary and secondary prevention trials assessing ICD and CRT devices.11-22) In fact, the mean patient age in these trials was 60 to 65 years, with few patients being >75 years of age. Recent studies suggest an increased mortality and a higher rate of procedural complications and worse outcomes in elderly patients undergoing device therapy,23 and therefore the risk-benefit ratio of ICD therapy and CRT may be skewed in older patients.
We will now review the current indications for ICD and CRT, clinical evidence for treatment, and unique features of elderly patients that should be considered when contemplating ICD therapy and CRT in this population.
Implantable Cardioverter-Defibrillator Therapy for Secondary Prevention of Sudden Cardiac Death
Three large, randomized controlled trials demonstrated a significant relative risk (RR) reduction in all-cause mortality for ICD therapy in patients who survived an episode of sustained ventricular tachycardia or ventricular fibrillation18-20; however, the elderly population was largely underrepresented in all of these trials. For example, only 3% of the 288 patients enrolled in the CASH (Cardiac Arrest Study Hamburg) trial20 were >75 years of age. Although the study found improved mortality trends with the use of ICD, there was no subgroup analysis to evaluate a potential age-dependent benefit of this therapy. In contrast, the AVID (Antiarrhythmics Versus Implantable Defibrillators) trial18 found a similar prognostic benefit with ICD implantation regardless of patient age in a prespecified subgroup analysis of individuals (<60 years vs 60-69 years vs ≥70 years). The CIDS (Canadian Implantable Defibrillator Study)19 investigators also identified a subgroup of older patients who were most likely to benefit from ICD therapy. Patients who met two of the following three parameters (age ≥70 years, left ventricular [LV] ejection fraction ≤35%, and New York Heart Association [NYHA] class III or IV) received greater benefit when compared with younger patients (<70 years of age) with better ejection fractions and a lower NYHA class (NYHA class <III).
An age-specific meta-analysis of the aforementioned trials was conducted to compare outcomes for older versus younger patients (1614 patients <75 years old vs 252 patients ≥75 years old).24 It was found that elderly patients ≥75 years of age had an increased risk of nonarrhythmic deaths and therefore did not derive the same benefit from ICD therapy seen in younger patients (hazard ratio [HR], 0.69; 95% confidence interval [CI], 0.56-0.85; P<.0001 for patients <75 years vs HR, 1.06; 95% CI, 0.69-1.64; P=.79 for patients ≥75 years). These results, however, are derived from meta-analysis and lack significant statistical power. Furthermore, they do not agree with the results of the prespecified group analysis of the AVID and CIDS trials.18,19 Therefore, based on the available data, ICD therapy for secondary prevention of sudden cardiac death should not be withheld in elderly patients based on age alone.
Implantable Cardioverter-Defibrillator Therapy for Primary Prevention of Sudden Cardiac Death
The MADIT-II (Multicenter Automatic Defibrillator Implantation Trial II)12 and the SCD-HeFT (Sudden Cardiac Death in Heart Failure Trial)14 are two of the most important primary prevention trials for ICD implantation. As with the secondary prevention trials discussed previously, the mean patient age in these studies was <65 years of age. SCD-HeFT is the largest primary prevention ICD study to date, enrolling 2521 patients with systolic LV dysfunction (ejection fraction ≤35% despite maximal medical therapy) to receive amiodarone, placebo, or an ICD in a randomized controlled fashion.14 The study population included patients with both ischemic and nonischemic cardiomyopathy. The mean age of the study population was 60 years. SCD-HeFT showed an absolute decrease in all-cause mortality of 7.2% after 5 years of follow-up for ICD recipients when compared with the amiodarone and placebo groups. Amiodarone showed a similar risk of death as placebo. A prespecified age subgroup analysis found a slightly higher mortality benefit for ICD versus placebo in 1098 patients <65 years of age (RR, 0.68; 95% CI, 0.5-0.9) when compared with 578 patients ≥65 years of age (RR, 0.86; 95% CI, 0.62-1.18).14 These results, however, lacked multivariate analysis for potential confounding comorbidities in elderly individuals and, therefore, the true effect of age on this SCD-HeFT population remains unclear. Another important finding from this study is the fact that older patients (≥65 years) who received amiodarone did not have an improved outcome (RR, 1.13; 95% CI, 0.8-1.5).14
In contrast with SCD-HeFT, MADIT-II exclusively enrolled patients with ischemic cardiomyopathy, defined as a history of remote myocardial infarction with an ejection fraction of ≤30% despite optimal medical therapy. A total of 1232 patients, including 204 patients ≥75 years of age, were enrolled. The main finding of this study, after a mean follow-up of 20 months, was a 31% RR reduction in all-cause mortality in the ICD group compared with the group receiving standard medical therapy alone.12 A subsequent subgroup analysis of this trial comparing patients ≥75 years of age with patients <75 years of age demonstrated a 46% RR reduction in total mortality in favor of the older patient population (P<.04).25
Based on the current evidence and practice guidelines, ICD therapy for primary prevention of sudden cardiac death should be considered in elderly patients with heart failure who have a reasonable life expectancy and no additional severe comorbidities.6
Cardiac Resynchronization Therapy
A significant proportion of patients with heart failure have evidence of ventricular dysynchrony, traditionally assessed by electrocardiography (QRS duration >120 ms) and/or echocardiography (significant delay in the contraction between the left and right ventricles). The choice of device (pacemaker or ICD) incorporating resynchronization therapy (known as CRT-P for pacemakers and CRT-D for defibrillators) to be implanted will depend on patient characteristics and specific indications for each particular device.
In addition to demonstrating a mortality benefit, multiple clinical trials have demonstrated the benefit of CRT in improving the symptoms of heart failure, quality of life, LV function, and exercise capacity21,22,26,27 in CRT recipients with advanced heart failure.21,22 Prespecified, age-related subgroup analysis of these studies demonstrated benefit for both young and older patients.21,22 Additionally, observational studies have also shown that the benefit of CRT extends to older patients. In a trial of 170 patients with NYHA class III and IV heart failure undergoing CRT, there was no significant difference in mortality or clinical outcomes (NYHA class, quality of life, exercise capacity, ejection fraction, and LV remodeling) between patients <70 years of age and ≥70 years of age during a 6-month follow-up period.28 Another prospective, observational study that enrolled 266 patients undergoing CRT implantation for advanced heart failure found no difference in mortality, clinical outcomes, or echocardiographic parameters when comparing patients <75 years of age with patients >75 years of age.29
Based on these studies, it appears that CRT provides significant improvement in quality of life and exercise capacity in elderly patients, including in those >80 years of age with advanced heart failure and dysynchrony with a left bundle branch block. Several questions still remain, however, regarding elderly patients and CRT that are not answered in current studies. Do elderly patients with atrial fibrillation and those with less advanced forms of heart failure (NYHA class II) benefit from CRT? How should comorbidities and patient preference influence the choice of CRT device (ie, CRT-P vs CRT-D)? Is the mortality benefit obtained with CRT-D greater than the risk imposed by concomitant illnesses in elderly individuals?
Particular Issues With Implantable Cardioverter-Defibrillator Therapy and Cardiac Resynchronization Therapy Devices in Older Patients
In a prospective study of 500 patients undergoing ICD therapy, long-term complication rates after 4 years of follow-up were similar in patients ≥75 years of age as in those <75 years of age (25% for older vs 23% for younger individuals; P=NS).30 The most frequent complications were inappropriate ICD shocks in 62 patients (12%), most commonly for atrial fibrillation with rapid ventricular response. In addition to the complications related to ICD therapy, CRT commonly requires the use of contrast dye injected directly into the coronary sinus in order to visualize suitable branches for an LV lead implantation. In elderly patients with or at high risk for acute renal failure, the risk of contrast-related nephrotoxicity must be considered. In our center, we often initially attempt implantation of an LV lead without the use of contrast dye in such patients, by selective cannulation of LV venous branches using wires and sheaths guided by fluoroscopy only. However, the likelihood of successful implantation with this technique is lower and we will therefore often need to perform contrast venography.
Although age was not a predictor of quality-of-life outcomes for ICD and CRT patients in all of the prospective major ICD and CRT trials, elderly individuals with multiple comorbidities were largely underrepresented (Table11-22). The benefits versus the risks of ICD and CRT must be discussed and considered individually for each elderly patient being considered for such device therapy. Also, the cost-effectiveness of device therapy has not been evaluated in large cohorts of elderly patients with heart failure who are recipients of ICD and CRT. To date, it remains unclear if the cost-benefit ratio of a younger patient population represented in the clinical trials is applicable to older individuals with coexisting comorbidities. Finally, older individuals who receive ICD or CRT devices and their families are more likely to face the issue of deactivation of a device in the event of end-stage heart failure refractory to medical therapy, ventricular tachycardia storm, or other terminal illnesses. In this situation, the deactivation of defibrillator therapies needs to be discussed with the patient and his or her family in order to avoid painful ICD shocks caused by incessant arrhythmias during the dying process.31,32
Conclusion
Device-based therapy of cardiac arrhythmias is commonly indicated in the elderly population. Although elderly individuals have been largely underrepresented in studies of these devices, pacemakers have been shown to have clear benefits in appropriately selected elders. Regarding ICDs, the data suggest that, as with younger individuals at increased risk of sudden cardiac death from severe LV dysfunction, sustained ventricular tachyarrhythmias, or aborted sudden cardiac arrest, older patients with heart failure who meet these criteria also experience a survival benefit. Additionally, CRT-P and CRT-D devices have demonstrated improvement in symptoms, exercise capacity, and prognosis in patients with heart failure and advanced intraventricular conduction delay. Due to a small number of patients >75 years of age participating in these studies, the risk-benefit ratio of defibrillator therapies in octogenarians is less clear. Current data from trials on modern ICD device implantation and endocardial leads show low perioperative mortality in selected patients with heart failure who are >75 years of age.33 Randomized trials and cohort studies have consistently shown benefit for elderly recipients of ICD and CRT devices, and therefore these therapies should not be withheld based on age alone.
Standard indications for the use of pacemaker, ICD, and CRT devices published in current guidelines are applicable to elderly patients, but in this population it becomes increasingly important to individualize therapy and weigh the risks versus the benefits, especially for those patients with significant comorbidities that place them at an increased risk of short-term, nonarrhythmic death. The decision to undergo a device implantation should be a collaborative effort between the physician, the patient, and his or her family to address the patient’s expectations and identify the indications, risks, and likely outcomes of device-based therapy. n
Dr. Saliaris is Assistant Professor, Department of Medicine, Division of Cardiology, and Dr. Jiménez is Electrophysiology Fellow, Adult Cardiac Electrophysiology Laboratories, University of Maryland Medical Center, Baltimore.
Dr. Saliaris has received speaker honoraria from and was a consultant for Medtronic. Dr. Jiménez reports no relevant financial relationships.
References
1. Masoudi FA, Havranek EP, Wolfe P, et al. Most hospitalized older persons do not meet the enrollment criteria for clinical trials in heart failure. Am Heart J. 2003;146(2):250-257.
2. Hall MJ, DeFrances CJ, Williams SN, Golosinskiy A, Schwartzman A. National Hospital Discharge Survey: 2007 summary. Natl Health Stat Report. 2010;26(29):1-20, 24.
3. Cheitlin MD, Gerstenblith G, Hazzard WR, et al. Database Conference January 27-30, 2000, Washington D.C.—Do existing databases answer clinical questions about geriatric cardiovascular disease and stroke? Am J Geriatr Cardiol. 2001;10(4):207-223.
4. Lamas GA, Orav EJ, Stambler BS, et al; Pacemaker Selection in the Elderly Investigators. Quality of life and clinical outcomes in elderly patients treated with ventricular pacing as compared with dual-chamber pacing. N Engl J Med. 1998;338(16):1097-1104.
5. Lopez-Jimenez F, Goldman L, Orav EJ, et al. Health values before and after pacemaker implantation. Am Heart J. 2002;144(4):687-692.
6. Epstein AE, DiMarco JP, Ellenbogen KA, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the ACC/AHA/NASPE 2002 Guideline Update for Implantation of Cardiac Pacemakers and Antiarrhythmia Devices); American Association for Thoracic Surgery; Society of Thoracic Surgeons. ACC/AHA/HRS 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the ACC/AHA/NASPE 2002 Guideline Update for Implantation of Cardiac Pacemakers and Antiarrhythmia Devices) developed in collaboration with the American Association for Thoracic Surgery and Society of Thoracic Surgeons [published corrections appear in J Am Coll Cardiol. 2009;53(16):1473 and J Am Coll Cardiol. 2009;53(1):147]. J Am Coll Cardiol. 2008;51(21):e1-e62.
7. Dhingra RC, Denes P, Wu D, Chuquimia R, Rosen KM. The significance of second degree atrioventricular block and bundle branch block. Observations regarding site and type of block. Circulation. 1974;49(4):638-646.
8. Langendorf R, Pick A. Atrioventricular block, type II (Mobitz)—its nature and clinical significance. Circulation. 1968;38(5):819-821.
9. Kenny RA, Richardson DA, Steen N, Bexton RS, Shaw FE, Bond J. Carotid sinus syndrome: a modifiable risk factor for nonaccidental falls in older adults (SAFE PACE). J Am Coll Cardiol. 2001;38(5):1491-1496.
10. Epstein AE, Kay GN, Plumb VJ, et al; ACT Investigators. Implantable cardioverter-defibrillator prescription in the elderly. Heart Rhythm. 2009;6(8):1136-1143.
11. Buxton AE, Lee KL, Fisher JD, Josephson ME, Prystowsky EN, Hafley G; Multicenter Unsustained Tachycardia Trial Investigators. A randomized study of the prevention of sudden death in patients with coronary artery disease [published correction appears in N Engl J Med. 2000;342(17):1300]. N Engl J Med. 1999;341(25):1882-1890.
12. Moss AJ, Zareba W, Hall WJ, et al; Multicenter Automatic Defibrillator Implantation Trial II Investigators. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346(12):877-883.
13. Hohnloser SH, Kuck KH, Dorian P, et al; DINAMIT Investigators. Prophylactic use of an implantable cardioverter-defibrillator after acute myocardial infarction. N EngI J Med. 2004;351(24):2481-2488.
14. Bardy GH, Lee KL, Mark DB, et al; Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT) Investigators. Amiodarone or an implantable cardioverter defibrillator for congestive heart failure [published correction appears in N Engl J Med. 2005;525(20):2146]. N Engl J Med. 2005;352(3):225-237.
15. Kadish A, Dyer A, Daubert JP, et al; Defibrillators in Non-Ischemic Cardiomyopathy Treatment Evaluation (DEFINITE) Investigators. Prophylactic defibrillator implantation in patients with nonischemic dilated cardiomyopathy. N Engl J Med. 2004;350(21):2151-2158.
16. Moss AJ, Hall WJ, Cannom DS, et al; Multicenter Automatic Defibrillator Implantation Trial Investigators. Improved survival with an implanted defibrillator in patients with coronary disease at high risk for ventricular arrhythmia. N Engl J Med. 1996;335(26):1933-1940.
17. Bigger JT Jr; Coronary Artery Bypass Graft (CABG) Patch Trial Investigators. Prophylactic use of implanted cardiac defibrillators in patients at high risk for ventricular arrhythmias after coronary artery bypass graft surgery. N Engl J Med. 1997;337(22):1569-1575.
18. The Antiarrhythmics Versus Implantable Defibrillators Investigators (AVID). A comparison of antiarrhythmic-drug therapy with implantable defibrillators in patients resuscitated from near-fatal ventricular arrhythmias. N Engl J Med. 1997;337(22):
1576-1583.
19. Connolly SJ, Gent M, Roberts RS, et al. Canadian implantable defibrillator study (CIDS): a randomized trial of the implantable cardioverter defibrillator against amiodarone. Circulation. 2000;101(11):1297-1302.
20. Kuck KH, Cappato R, Siebels J, Rüppel R. Randomized comparison of antiarrhythmic drug therapy with implantable defibrillators in patients resuscitated from cardiac arrest: the Cardiac Arrest Study Hamburg (CASH). Circulation. 2000;102(7):748-754.
21. Bristow MR, Saxon LA, Boehmer J, et al; Comparison of Medical Therapy, Pacing and Defibrillation in Heart Failure (COMPANION) Investigators. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med. 2004;350(21):2140-2150.
22. Cleland JG, Daubert JC, Erdmann E, et al; Cardiac Resynchronization-Heart Failure (CARE HF) Study Investigators. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med. 2005;352(15):1539-1549.
23. Swindle JP, Rich MW, McCann P, Burroughs TE, Hauptman PJ. Implantable cardiac device procedures in older patients: use and in-hospital outcomes. Arch Intern Med. 2010;170(7):631-637.
24. Healey JS, Hallstrom AP, Kuck KH, et al. Role of the implantable defibrillator among elderly patients with a history of life-threatening ventricular arrhythmias. Eur Heart J. 2007;28(14):1746-1749.
25. Huang DT, Sesselberg HW, Salam T, McNitt S, Noyes K, Andrews M; MADIT-II Research Group. Improved survival associated with prophylactic implantable defibrillators in elderly patients with prior myocardial infarction and depressed ventricular function: a MADIT-II substudy. J Cardio Electrophysiol. 2007;18(8):833-838.
26. McAlister FA, Ezekowitz JA, Wiebe N, et al. Systematic review: cardiac resynchronization in patients with symptomatic heart failure [published correction appears in Ann Intern Med. 2005;142(4):311]. Ann Intern Med. 2004;141(5):381-390.
27. Young JB, Abraham WT, Smith AL, et al; Multicenter InSync ICD Randomized Clinical Evaluation (MIRACLE ICD) Trial Investigators. Combined cardiac resynchronization and implantable cardioversion defibrillation in advanced chronic heart failure: the MIRACLE ICD Trial. JAMA. 2003;289(20):2685-2694.
28. Bleeker GB, Schalij MJ, Molhoek SG, et al. Comparison of effectiveness of cardiac resynchronization therapy in patients <70 versus > or =70 years of age. Am J Cardiol. 2005;96(3):420-422.
29. Delnoy PP, Ottervanger JP, Luttikhuis HO, et al. Clinical response of cardiac resynchronization therapy in the elderly. Am Heart J. 2008;155(41):746-751.
30. Grimm W, Stula A, Sharkova J, Maisch B. Outcomes of elderly recipients of implantable cardioverter defibrillators [published correction appears in Pacing Clin Electrophysiol. 2007;30(11):1424]. Pacing Clin Electrophysiol. 2007;30(suppl 1):S34-S38.
31. Basta LL. End-of-life and other ethical issues related to pacemaker and defibrillator use in the elderly. Am J Geriatr Cardiol. 2006;15(2):114-117.
32. Goldstein NE, Lampert R, Bradley E, Lynn J, Krumholz HM. Management of implantable cardioverter defibrillators in end-of-life care. Ann Intern Med. 2004;141(11):835-838.
33. Kong MH, Al-Khatib SM, Sanders GD, Hasselblad V, Peterson ED. Use of implantable cardioverter-defibrillators for primary prevention in older patients: a systematic literature review and meta-analysis. Cardiol J. 2011;18( 5):503-514.


