A Collection of Cardiovascular Events
Takotsubo Cardiomyopathy
Ibrahim M. Elali, MD, and Jose C. Missri, MD
The acute onset of substernal chest pain during vigorous exercise prompted a 70-year-old woman to seek emergency medical attention. She reported chest pressure, left arm numbness, shortness of breath, and severe sweating.
The patient had mild chronic obstructive pulmonary disease, osteoporosis, and medically controlled ulcerative colitis. She had no history of cardiac symptoms. An ECG obtained during a complete physical examination performed 6 days before this event had appeared normal.
In the emergency department, the patient was in severe respiratory distress. She was afebrile and had a blood pressure of 87/70 mm Hg, heart rate of 88 beats per minute, respiration rate of 25 breaths per minute, and oxygen saturation of 83% while breathing room air. She had bilateral rales, normal heart sounds, and a grade 2/3 systolic murmur over the apex that radiated into the axilla.
Laboratory testing. Troponin T level was 0.89 µg/L (greater than 0.07 µg/L is considered abnormal); thyroid-stimulating hormone level was slightly decreased, with normal free thyroxine and total triiodothyronine levels; and white blood cell count was 23,500/µL, with 93.6% neutrophils. Remaining laboratory tests revealed no significant abnormalities. Findings from computed radiography were consistent with pulmonary edema. An ECG revealed normal sinus rhythm and segment elevation in V3-V5.
The preliminary diagnosis was acute anterior wall myocardial infarction (MI) with cardiogenic shock and pulmonary edema. The patient received tenecteplase, low-dose aspirin, and heparin. She was then transferred emergently to our facility for urgent cardiac catheterization and angioplasty.
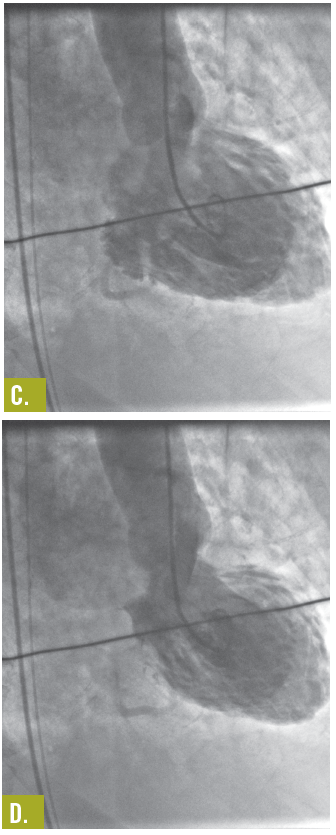
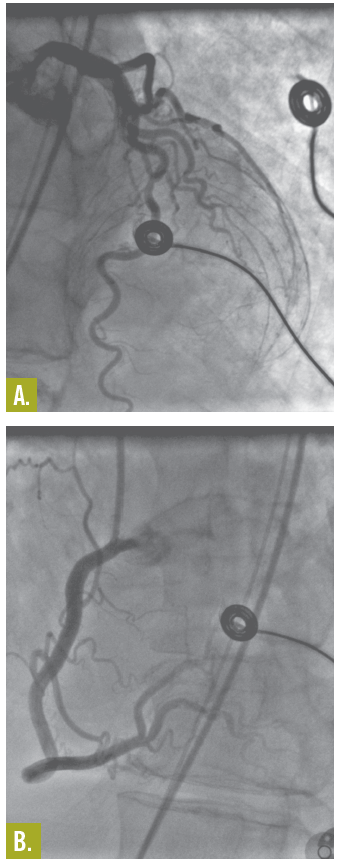
Despite thrombolytic therapy, the ECG did not demonstrate significant changes. Cardiac catheterization showed normal coronary arteries (Figures A and B), a left ventricular (LV) ejection fraction of 20%, and grade 2 to grade 4 mitral regurgitation. Findings on an aortogram were normal. A ventriculogram showed transient LV apical ballooning syndrome (also known as takotsubo cardiomyopathy [TC]) and akinesia (Figures C and D).
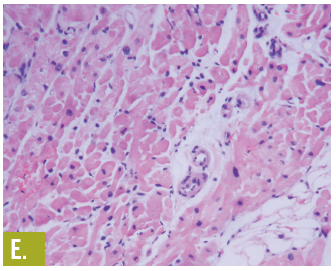
A myocardial biopsy specimen showed mild hypertrophy of the myocytes and a small amount of interstitial fat (Figure E). These nonspecific findings can be seen with cardiomyopathy of undetermined cause. A follow-up echocardiogram on day 22 showed an ejection fraction of 60% with normal-appearing valves and heart chamber. The patient continued to improve, and on day 23 was sent home to complete a cardiac rehabilitation program.
Discussion. TC is a transient and reversible LV dysfunction characterized by apical and midventricular dyskinesia with basal hyperkinesis, in response to an insult. The mechanism is yet to be identified and is most probably multifactorial. The most popular hypothesis is catecholaminergic or adrenoceptor-hyperactive cardiomyopathy,1,2 coupled with stress-induced transient coronary endothelial dysfunction,3,4 possibly aggravated by estrogen deficiency.5
TC mostly occurs in women older than 60 and is often preceded by emotional or physical stress. The diagnosis, as well as the findings, is usually determined from the ECG, coronary angiogram, and ventriculogram. It is made on the basis of the history and clinical presentation symptoms and presentation resemble those of acute MI. In TC, however, levels of myocardial serum markers are elevated, coronary arteries are disease-free, and the heart is shaped like a takotsubo (the Japanese word for a pot used to catch octopus).
Outcome of the case. TC generally improves rapidly within a few weeks with supportive management, with almost full recovery of LV function, although traditional heart failure medication has been used. As was the case in this patient, prognosis is very good, with low recurrence and mortality rate. ■
References:
1. Ueyama T, Kasamatsu K, Hano T, et al. Emotional stress induces transient left ventricular hypocontraction in the rat via activation of cardiac adrenoreceptors: a possible animal model of “tako-tsubo” cardiomyopathy. Circ J. 2002;66:712-713.
2. Mori H, Ishikawa S, Kojima S, et al. Increased responsiveness of left ventricular apical myocardium to adrenergic stimuli. Cardiovasc Res. 1993;27:192-198.
3. Ghiadoni L, Donald AE, Cropley M, et al. Mental stress induces transientendothelial dysfunction in humans. Circulation. 2000;102:
2473-2478.
4. Spieker LE, Hürlimann D, Ruschitzka F, et al. Mental stress induces prolonged endothelial dysfunction via endothelin-A receptors. Circulation. 2002;105:2817-2820.
5. Ueyama T, Hano T, Kasamatsu K, et al. Estrogen attenuates the emotional stress-induced cardiac responses in the animal model of Tako-tsubo (Ampulla) cardiomyopathy. J Cardiovasc Pharmacol. 2003;42(suppl 1):S117-S119.
Hypotension in a Man with Acute MI
William J. Brady, MD, Richard A. Harrigan, MD, Theodore Chan, MD
A 56-year-old man with a history of diabetes mellitus and hypertension presents with chest pain, emesis, pallor, and diaphoresis. The chest pain began the day before as intermittent, left-sided discomfort that subsequently became constant; over the past 2 hours, it has intensified.
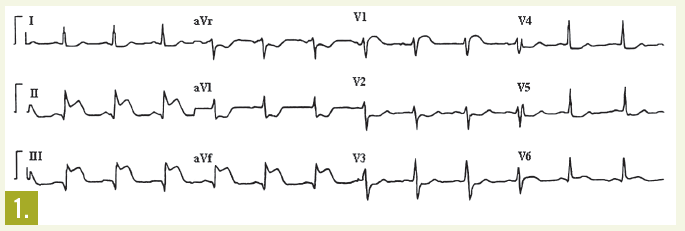
Physical examination. Blood pressure is 115/78 mm Hg; heart rate, 65 beats per minute; and respiration rate, 30 breaths per minute. Neck veins are markedly distended; lung fields are clear. A 12-lead ECG shows sinus rhythm with ST-segment elevation in leads II, III, and aVF; ST-segment depression is evident in leads I and aVL. These ECG findings are interpreted as acute inferior wall myocardial infarction (MI) with reciprocal ST-segment depression in the lateral leads.
The patient is given aspirin and sublingual nitroglycerin (0.4 mg) while an intravenous line is placed. Moments later, the patient vomits. Blood pressure is undetectable; heart rate is 90 beats per minute; ECG monitoring continues to show normal sinus rhythm. While intravenous saline is administered, a second ECG is obtained; it does not reveal interval change from the first. Then a third ECG, using right-sided precordial leads, is obtained.
This patient sustained an inferior wall MI with right ventricular infarction. The typical approach to diagnosis and management of acute coronary syndromes focuses on different anatomic segments of the left ventricle (eg, the inferior wall). Right ventricular MI is a distinct acute coronary syndrome.
Discussion. Right ventricular MI is defined as hypotension, elevated jugular venous pressure, and shock in the presence of clear lung fields in patients with acute coronary syndrome.1 Clinically recognized right ventricular MI usually occurs in the setting of an acute left ventricular ST-segment elevation MI (STEMI). Isolated right ventricular MI is rare.
In most patients with right ventricular MI, the inferior wall of the left ventricle is involved (usually in the form of a STEMI) as a result of occlusion of the right coronary artery proximal to the right ventricular branch. However, in a few patients (about 10% of those in whom right ventricular MI develops), the left circumflex artery supplies the right ventricle and a lateral wall STEMI occurs.
The incidence of right ventricular MI varies significantly, depending on the method used to detect such injury. ECG studies have consistently shown that right ventricular MI occurs in about one-third of patients with inferior wall STEMI.2-4 However, autopsy studies reveal that right ventricular MI occurs in more than half of patients with acute MI.
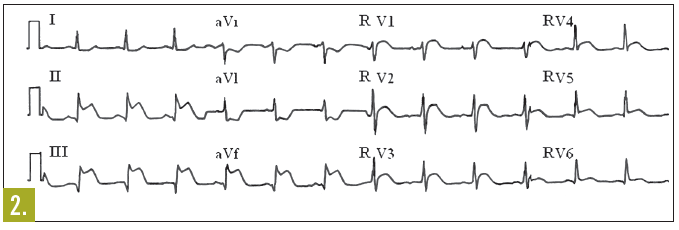
Laboratory testing. Several findings on the standard 12-lead ECG suggest right ventricular acute MI, including ST-segment elevation in the inferior leads (II, III, and aVF)3,5 and in the right precordial chest leads, particularly lead V1 (Figure 1). In fact, lead V1 is the only standard lead of the 12-lead ECG that directly images the right ventricle.6-8
On occasion, coexisting acute MI of the posterior wall of the left ventricle may obscure the ST-segment elevation in lead V1 that is caused by right ventricular MI; this occurs in patients who have acute inferoposterior MI with right ventricular involvement.9-11 However, a particular pattern of the relative magnitudes of the ST-segment elevation in the various inferior leads—greater ST-segment elevation in lead III than in the other inferior leads—suggests right ventricular acute MI. This pattern of ST-segment elevation occurs because lead III reflects the activity of the right ventricle more directly than do the other inferior leads.
Nonstandard electrocardiographic imaging can also be useful when right ventricular MI is suspected. Both additional ECG leads and body surface mapping have demonstrated varying degrees of success as diagnostic aids in this setting. The addition of lead RV4 to the standard 12-lead ECG provides further objective evidence of right ventricular involvement: right ventricular MI can be diagnosed with 80% to 100% sensitivity if ST-segment elevation of more than 1 mm is seen in this lead.3,12 All the right-sided leads, RV1 through RV6, show evidence of right ventricular MI; however, use of lead RV4 alone has yielded rates of right ventricular MI diagnosis similar to those obtained when the entire array of right-sided ECG leads was used (Figure 2).4
The ST-segment elevation in the right-sided leads is usually less pronounced than that seen on a standard 12-lead ECG in patients with left ventricular STEMI. This disparity results from the fact that the right ventricle is composed of considerably less muscle than the left ventricle; because there is less myocardium to manifest a current of injury, the ST segment is less elevated.
This recently developed—but not yet widely used—electrocardiographic technique involves placing numerous leads on a patient’s anterior and posterior chest, thereby enabling more complete visualization of cardiac electrical activity. Output from body surface mapping can be displayed in a 12-lead ECG format, in an 80-lead ECG format, and on color contour maps. The color contour maps can be displayed on a torso image or as flat maps. The body map approach to ECG imaging has increased the diagnosis of right ventricular MI in patients with acute coronary syndrome.13
Identifying right ventricular involvement is important because patients with coexisting inferior wall infarction have a large amount of jeopardized myocardium. Consequently, they are at increased risk for life-threatening complications, including high-grade atrioventricular block,3 atrial fibrillation, symptomatic sinus bradycardia, atrial infarction (signaled by PR-segment displacement, elevation, or depression in lead II, III, or aVF), cardiogenic shock, and cardiopulmonary arrest.12 In fact, the complication rate seen in patients with this type of infarction is similar to that seen in patients with anterior wall acute MI.10
Aggressive therapy is often warranted to limit adverse events. Mortality and in-hospital complications were significantly reduced in patients with right ventricular acute MI (diagnosed on the basis of ST-segment elevation in lead RV4) who received fibrinolysis.14 Also, because nitrate-induced hypotension has been well described in patients with right ventricular MI,15 ST-segment elevation in lead RV4 should prompt cautious use of nitrates and other vasodilators as well as judicious administration of crystalloid infusions to avoid systemic hypotension.
Outcome of the case. In the ECG with right-sided leads (Figure 2), ST-segment elevation was noted in leads RV1 through RV6; these findings are consistent with right ventricular MI in the setting of acute inferior wall MI. Further review of the initial 12-lead ECG revealed disproportionate ST-segment elevation in lead III.
After administration of 600 mL of normal saline, the patient’s blood pressure was detectable at 105/60 mm Hg. He was transferred to the cardiac catheterization laboratory, where a proximal right coronary artery lesion with occlusive thrombus was detected and a stent was successfully placed. Hypokinesis of the right ventricle and inferior wall of the left ventricle was also noted. Serum troponin levels were elevated, confirming the diagnosis of acute MI. He had an uneventful recovery; good left ventricular function was restored.
References:
1. Cohn JN, Guiha NH, Broder MI, Limas CJ. Right ventricular infarction. Clinical and hemodynamic features. Am J Cardiol. 1974;33:209-214.
2. Zeymer U, Neuhaus KL, Wegscheider K, et al. Effects of thrombolytic therapy in acute myocardial infarction with or without right ventricular involvement. HIT-4 Trial Group. Hirudin for Improvementof Thrombolysis. J Am Col Cardiol. 1998;32:876-881.
3. Braat SH, Brugada P, de Zwaan C, et al. Value of electrocardiogram in diagnosing right ventricular involvement in patients with an acute inferior wall myocardial infarction. Br Heart J. 1983;49:368-372.
4. Klein HO, Tordjman T, Ninio R, et al. The early recognition of right ventricular infarction: diagnostic accuracy of the electrocardiographic V4R lead. Circulation.1983;67:558-565.
5. Andersen HR, Nielsen D, Falk E. Right ventricular infarction: diagnostic value of ST elevation in lead III exceeding that of lead II during inferior/posterior infarction and comparison with right-chest leads V3R to V7R. Am Heart J. 1989;117:82-86.
6. Lopez-Sendon J, Coma-Canella I, Alcasena S, et al. Electrocardiographic findings in acute right ventricular infarction: sensitivity and specificity of electrocardiographic alterations in right precordial leads V4R, V3R, V1, V2, and V3. J Am Coll Cardiol. 1985;6:1273-1279.
7. Croft CH, Nicod P, Corbett JR, et al. Detection of acute right ventricular infarction by right precordial electrocardiography. Am J Cardiol. 1982;50:421-427.
8. Coma-Canella I, Lopez-Sendon J, Alcasena S, et al. Electrocardiographic alterations in leads V1 to V3in the diagnosis of right and left ventricular infarction. Am Heart J. 1986;112:940-946.
9. Zalenski RJ, Cooke D, Rydman R, et al. Assessing the diagnostic value of an ECG containing leads V4R, V8, and V9: the 15-lead ECG. Ann Emerg Med. 1993;22:786-793.
10. Zalenski RJ, Rydman RJ, Sloan EP, et al. Value of posterior and right ventricular leads in comparisonto the standard 12-lead electrocardiogram in evaluation of ST-segment elevation in suspectedacute myocardial infarction. Am J Cardiol. 1997;79:1579-1585.
11. Aufderheide TP, Brady WJ. Electrocardiography in the patient in myocardial ischemia or infarction. In: Gibler WB, Aufderheide TP, eds. Emergency Cardiac Care. St Louis, MO: Mosby, Inc; 1994:169-216.
12. Haji SA, Movahed A. Right ventricular infarction—diagnosis and treatment. Clin Cardiol. 2000;23:473-482.
13. Self WH, Mattu A, Martin M, et al. Body surface mapping in the ED evaluation of the patient with chest pain: use of the 80-lead electrocardiogram system. Am J Emerg Med. 2006;24:87-112.
14. Zehender M, Kasper W, Kauder E, et al. Eligibility for and benefit of thrombolytic therapy in inferior myocardial infarction: focus on the prognostic importance of right ventricular infarction. J Am Coll Cardiol. 1994;24:362-369.
15. Brady WJ, Hwang V, Sullivan R, et al. A comparison of 12- and 15-lead ECGs in ED chest pain patients: impact on diagnosis, therapy, and disposition. Am J Emerg Med. 2000;18:239-243.
Young Man with Severe Hypertension and Palpitations
Navin M. Amin, MD
A 22-year-old man is hospitalized because of severe hypertension. In addition, he reports exertional retrosternal pressure-like pain, with a severity of 5/10, which lasts for 2 to 3 minutes. The pain does not radiate and is relieved with rest; there is no nausea, vomiting, diaphoresis, dizziness, or syncope. Two years earlier, he noticed dyspnea after a half-block walk on level ground; he also experienced intermittent episodes of palpitations.
The patient denies paroxysmal nocturnal dyspnea, orthopnea, ankle edema, and claudication in the lower legs. He has no history of headaches, vision problems, weakness, paresthesia, confusion, seizures, syncope, cough, hemoptysis, weight change, or urinary symptoms. There is no rash, adenopathy, diarrhea, abdominal pain, or vomiting.
The patient does not smoke, drink alcohol, or use illicit drugs. He is allergic to sulfa drugs.
Physical examination. The patient, who appears anxious, is well-built and well-nourished. He is 66 inches tall and weighs 120 lbs. He is afebrile. His heart rate is 76 beats per minute and regular; his respiration rate is 22 breaths per minute. Blood pressure (BP) in right upper limb, 210/122 mm Hg; left upper limb, 208/120 mm Hg; and right lower limb, 120/72 mm Hg. Hydration status is good. Examination of the head and neck reveals no icterus, erythema, or candidal infection. There is no evidence of anemia, clubbing, or cyanosis. The thyroid is not palpable. No ankle edema or tattoos are noted.
Jugular vein pulse and heart sounds are normal. Peripheral pulses in the lower limbs, particularly the femoral arteries, manifest low volume and delayed upstroke, compared with peripheral pulses in the upper limbs and carotid arteries. There is pulsation in the suprasternal notch. The apex is in the sixth intercostal space, outside the midclavicular line, on the left side. A grade 2 systolic murmur is audible in the interscapular region. Lungs are clear. An abdominal exam reveals no organomegaly, tenderness, or bruits. No striae are seen. The results of neurologic and funduscopic exams are normal.
Laboratory testing. White blood cell count, 5100/µL, with normal differential; hemoglobin level, 15.7 g/dL; hematocrit value, 47%; platelet count, 238,000/µL; erythrocyte sedimentation rate, 12 mm/h. Urinalysis results are normal. Serum sodium level is 139 mEq/L; potassium, 4.5 mEq/L; chloride, 102 mEq/L; carbon dioxide, 24 mEq/L; glucose, 95 mg/dL. Blood urea nitrogen level, 14 mg/dL; creatinine, 0.8 mg/dL; calcium, 9.5 mg/dL; total serum bilirubin, 1 mg/dL; alkaline phosphate, 96 U/L. Total protein level, 8 g/dL; albumin, 4.6 g/dL; aspartate aminotransferase, 26 U/L; alanine aminotransferase, 32 U/L. An ECG shows left atrial enlargement. An echocardiogram reveals an ejection fraction of 60% and mild concentric left ventricular (LV) hypertrophy. Chest films are ordered.
Discussion. In patients who have a coarctation, the narrowing of the lumen of the aorta obstructs blood flow and thereby raises BP. This relatively common anomaly accounts for 5% to 8% of congenital cardiovascular defects. Coarctation may occur in isolation or in association with other cardiac abnormalities, such as bicuspid aortic valve or ventricular septal defect, or with Turner or DiGeorge syndrome.
Physical examination reveals hypertension in the upper extremities. A systolic murmur over the left precardium or between the scapulae may be present, and the femoral pulses may be absent or diminished, with delayed upstroke. Enlarged palpable collateral vessels with audible bruits may be observed at the scapular margin.
Diagnostic studies. The ECG findings are usually normal but may show LV hypertrophy in severe coarctation. Echocardiography is useful in assessing LV function. Two-dimensional echocardiography can delineate the site of coarctation and indicate associated cardiac anomalies. Color Doppler ultrasonography can suggest the magnitude of pressure gradients.
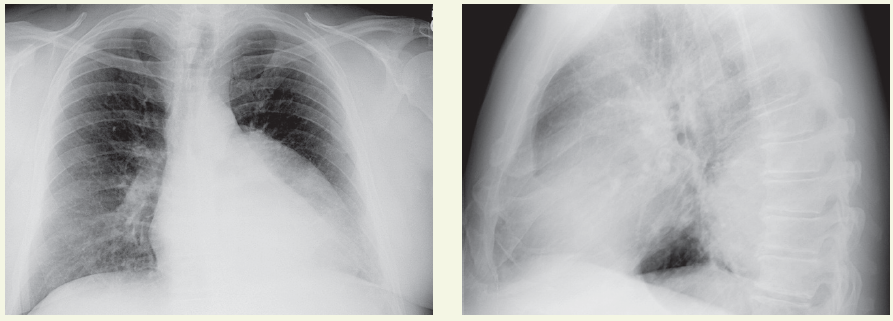
Chest radiography may reveal LV hypertrophy, the classic sign of rib notching (which is caused by erosion of the lower edges of the ribs by dilated intercostal arteries), and the “reverse 3” sign related to the aortic arch and descending aorta. The upper part of the 3 is formed by the dilated proximal segment descending into the coarctated segment. The bottom portion of the 3 is formed by the coarctated segment as it exits into the normal distal segment of the aorta.
Chest CT and MRI clearly reveal the dilated, narrowed segment and collaterals, as well as other abnormalities. However, angiography and aortography are the gold-standard studies to pinpoint the exact location and extent of the coarctation, delineate great-vessel involvement, facilitate evaluation of associated cardiac defects, and allow direct measurement of pressure gradients.
Treatment. Medical therapy is limited to optimization of hemodynamics before surgery. Prostaglandin E1 therapy and aggressive measures to control congestive heart failure are undertaken in infants. Surgery is the definitive treatment. Options include end-to-end reanastomosis, subclavian flap aortoplasty, prosthetic patch onlay grafts, and interposition grafts. The role of balloon angioplasty of native coarctation remains controversial. ■


