A Collection of Cardiometabolic Syndromes
Wellens Syndrome
Zaki A. Refaat, MD, and Stephen M. Fleet, MD
A 72-year-old obese man with chronic atrial fibrillation, hypertension, hyperlipidemia, and a history of tobacco use presented for a routine office visit. A year earlier, he began to experience recurrent chest pain, but an ECG had shown normal T waves. An adenosine pharmacological stress test with single photon emission CT myocardial perfusion imaging showed borderline ST changes, which were nondiagnostic. Nuclear images were negative for ischemia, and the ejection fraction was 52%.
Chest pain recurred intermittently for several months during rest and exertion. At the time of his office visit, however, the patient was entirely asymptomatic. Findings from a physical examination of the lungs, heart, abdomen, extremities, and pulses were unremarkable.
Office ECG findings showed atrial fibrillation with a controlled heart rate. Deep T-wave inversions were noted across the precordium (A), however. The patient was admitted to the hospital. Serial creatine kinase and troponin levels were normal. Warfarin was discontinued and cardiac catheterization was performed once the patient’s international normalized ratio was normal.
Coronary arteriography revealed single-vessel disease with a 90% stenosis of the proximal left anterior descending (LAD) artery (B). Ejection fraction was 50%. The patient received a drug-eluting stent to the LAD artery lesion and was discharged home the following day in good condition.

Wellens syndrome suggests severe disease of the proximal LAD artery. Classic ECG findings are characterized by deep (greater than 2 mm) symmetric T-wave inversions in the anterior precordial leads, usually in the setting of unstable angina.1,2 The ECG manifestations may occur on or after hospital admission and during pain-free intervals.1-3 Despite the absence of ST-segment elevation or a significant rise in cardiac enzyme levels, the characteristic Wellens syndrome ECG suggests tight proximal LAD coronary artery stenosis, which carries a poor prognosis if it is unrecognized or managed medically or if the patient is sent for diagnostic stress testing.1-3 Variant ECG findings in the anterior precordial leads include biphasic T waves and combined ST depression and T-wave inversion.3,4
If Wellens syndrome is recognized in a timely fashion, appropriate evaluation and treatment will avert an otherwise poor prognosis. Patients with this ECG finding should undergo cardiac catheterization rather than stress testing, so as not to precipitate an acute cardiac event in the stress laboratory.
Emphysematous Pyelonephritis
Tenecia Deans, MD, and Stacy Higgins, MD
For 4 days, a 62-year-old woman with type 2 diabetes mellitus had constant, severe, crampy abdominal pain associated with nausea, nonbloody emesis, and anorexia, but not fever. She reported that the pain had an insidious onset and was exacerbated by urination. Dysuria began 2 weeks before the onset of pain.
Physical examination. The patient’s temperature was 35.4°C (95.7°F), blood pressure was 105/61 mm Hg, pulse was 122 beats per minute, respiration rate was 24 breaths per minute, and glucose level was 450 mg/dL. The patient was awake, alert, and appropriately oriented. The physical examination was notable for a soft, obese abdomen with mild bilateral upper quadrant tenderness on deep palpation.
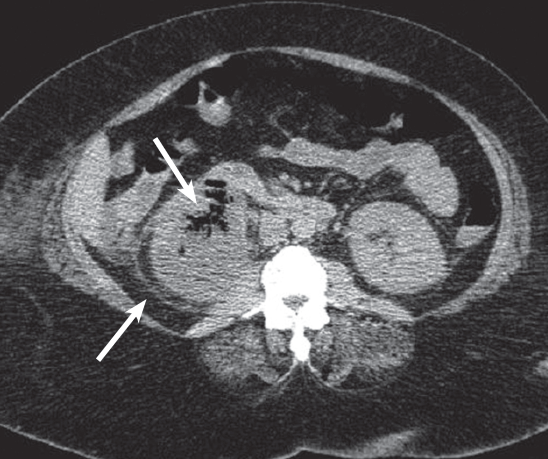
Laboratory testing. White blood cell count was 17,300/µL with 86% neutrophils and 9% bands, platelet count was 157,000/µL, blood urea nitrogen level was 64 mg/dL, and creatinine level was 5 mg/dL. Urinalysis showed 3+ proteinuria, glycosuria, positive leukocyte esterase, as well as pyuria and bacteria. A CT scan of the abdomen (Figure) revealed an enlarged, nonhydronephrotic right kidney with perinephric stranding, gas within the cortex (thick arrow) and perinephric spaces (thin arrow), as well as inflammation and stranding around the right proximal ureter.
The patient was admitted to the ICU for management of emphysematous pyelonephritis (EPN). Her ICU course was notable for the development of diabetic ketoacidosis and septic shock. Pressors, broad-spectrum antibiotics, and insulin were initiated. Blood and urine cultures eventually grew Escherichia coli. Her hospital stay was complicated by the development of oliguric renal failure, which required hemodialysis.
Clinical findings with significant negative prognostic value in EPN are thrombocytopenia, altered mental status, acute renal failure, and shock. Factors such as age, blood glucose level, and site of infection do not have a significant influence on outcome. Characteristics that best predict a favorable outcome are nonobstructive unilateral disease, combined medical and surgical treatment, and short interval between symptom onset and initiation of therapy.1
Discussion. EPN is usually diagnosed by CT and classified based on imaging findings2:
• Class 1: gas in the collecting system only.
• Class 2: gas in the renal parenchyma without extension to the extrarenal space.
• Class 3A: extension of gas or abscess to the perinephric space.
• Class 3B: extension of gas or abscess to the pararenal space.
• Class 4: bilateral EPN or a solitary kidney with disease.
For classes 1 and 2, combined antibiotic therapy and percutaneous nephrostomy tube drainage (PCD) have a success rate of 66%, while the mortality rate for treatment with antibiotics alone ranges from 40% to 90%.1,3
For classes 3 and 4, with 2 or fewer negative prognostic indicators, antibiotic therapy and PCD may be attempted but ultimately nephrectomy may be necessary.1,4 With more than 2 negative prognostic indicators, nephrectomy is recommended. The success rate of nephrectomy was 90% when attempted in 1 study.1
Given that this patient’s EPN was class 3B, with 2 negative prognostic indicators, nephrectomy was considered because of her critical condition; however, she instead underwent PCD and antibiotic therapy for kidney preservation.
EPN should be suspected in patients with diabetes and/or urinary tract
obstruction with acute renal failure and E coli or Klebsiella pneumoniae infection. Although EPN is rare, the projected growth of the diabetic population warrants increased attention to this potentially fatal disease.
Outcome of the case. Serial CT scans showed substantial improvement. She was discharged home on ciprofloxacin and scheduled for continued hemodialysis.
References:
1. Huang J, Tseng C. Emphysematous pyelonephritis. Arch Intern Med. 2000;160(6):797-805.
2. Mallet M, Knockaert D, Oyen R, Van Poppel H. Emphysematous pyelonephritis: no longer a surgical disease? Eur J Emerg Med. 2002;9(3):266-269.
3. Wan Y, Lee T, Bullard M, Tsai C. Acute gas-producing bacterial renal infection: correlation between imaging findings and clinical outcome. Radiology. 1996;
198(2):433-438.
4. Rasoul M, Keyvan R. Emphysematous urinary tract infections: diagnosis, treatment, and survival (case review series). Am J Med Sci. 2007;333(2):111-116.
Calciphylaxis
Raja Shekhar, R. Sappati Biyyani, MD, and Salim M. Saiyed, MD
A 55-year-old woman was hospitalized with pain and ulcers on the right breast, fourth digit of the right hand, and left fifth toe. She had hypertension, end-stage renal disease (ESRD) for which she was receiving hemodialysis, atrial fibrillation, and coronary artery disease, and she was morbidly obese (body mass index [BMI] of 45 kg/m2). Medications included aspirin, metoprolol, atorvastatin, sevelamer, and isosorbide mononitrate. The patient denied using tobacco, alcohol, or illicit drugs. For the past year, she had been taking warfarin only intermittently and had discontinued it altogether a month before presentation.
History. About 10 days earlier, painful hyperpigmented, violaceous, and indurated plaque-like ulcers with central eschar developed on the patient’s right breast. She had fever and chills while undergoing dialysis, which prompted the hospital admission.
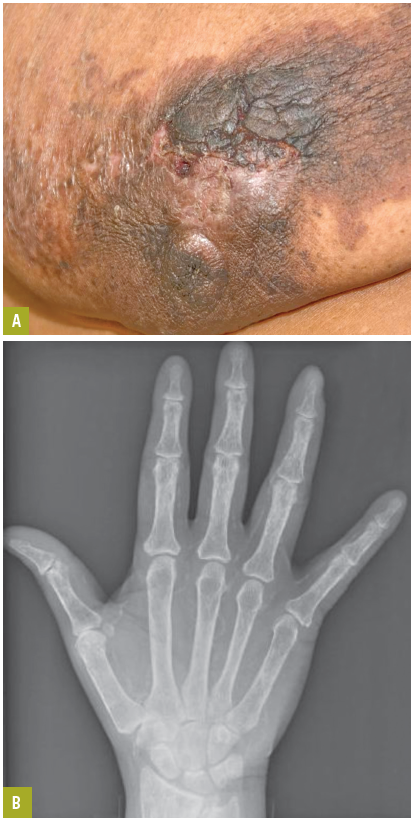
On examination, the patient was afebrile; vital signs were normal. Erythematous 4 cm x 3 cm lesions with induration and a surrounding area of 10 cm x 8 cm erythema were noted on the right breast (Figure A). Similar but smaller lesions were noted on the right fourth digit of the hand and left fifth toe. Abnormal laboratory values were as follows: calcium at 10.1 mg/dL (normal range, 8.5-10.5 mg/dL), phosphorus at 7.9 mg/dL (normal, 2.6-4.5 mg/dL), albumin at 3.1 g/dL (normal, 3.3-5 g/dL), and intact parathyroid hormone at 2280 pg/mL (normal, 10-55 pg/mL).
Electrocardiographic findings showed atrial fibrillation with a rapid ventricular rate of 160 beats per minute with no ischemic changes. Heart rate was controlled with beta-blockers, and myocardial ischemia was ruled out with negative cardiac enzymes. Chest radiograph showed atherosclerotic calcifications of the aortic knob and cardiomegaly. Radiographs of the patient’s hand showed extensive atherosclerotic vascular calcifications (Figure B). Arterial Doppler ultrasonography of the extremities showed right femoral, left ileo-femoral, and right radial and ulnar artery occlusive disease with mild distal ischemia at rest.
Because the patient had no predisposing cardiac conditions and blood cultures were negative, bacterial endocarditis was ruled out. Autoimmune serology was negative. Clinical presentation, high calcium-phosphate product, secondary hyperparathyroidism, and radiographic evidence of vascular calcification were highly suggestive of calciphylaxis. Intravenous fluids and calcitonin were added to the drug regimen, and the sevela-mer dosage was increased from 800 mg to 1600 mg, 3 times a day.
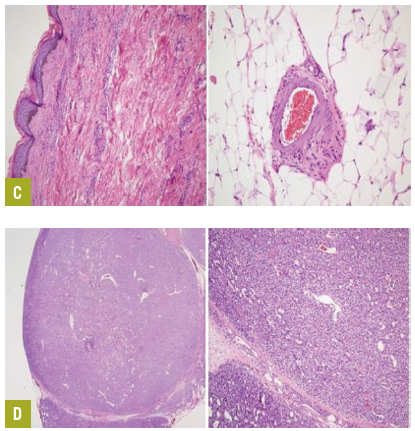
Laboratory testing. Punch biopsy results of right breast lesions were inconclusive. A subsequent deep tissue biopsy results displayed superficial dermal angiomatosis with chronic inflammation and abundant adipose tissue (Figure C). Technitium-sestamibi scan suggested a left inferior parathyroid adenoma. The patient underwent successful emergent subtotal parathyroidectomy for severe secondary hyperparathyroidism.
An examination of a biopsy specimen stained with hematoxylin and eosin demonstrated hyperplasia with dystrophic calcification (Figure D). After surgery, the calcium-phosphate product was 20 and parathyroid hormone level 3.8 pg/mL. Pain and skin lesions slowly seemed to be resolving.
Discussion. Calciphylaxis is a rare phenomenon with an estimated incidence of 1% to 4.5% among dialysis patients.1 It was first described as a hypersensitivity reaction after exposure to certain “sensitizers” by Seyle in 1962.2 Small artery medial calcification with endovascular fibrosis leading to low perfusion pressures and the activation of inflammatory mediators with hemodialysis cause endothelial damage resulting in ischemic skin lesions. Distal extremities are involved in 90% of cases. Involvement of breast tissue, trunk, and proximal extremities carries a poorer prognosis.3 Calciphylactic lesions are nonhealing and prone to secondary bacterial infections. Mortality approaches 80% without treatment. Systemic calciphylaxis can result in painful ischemic myopathy with muscle weakness and/or severe myositis.4
Risk factors for calciphylaxis include obesity, female gender, Caucasian race, low albumin levels, elevated serum alkaline phosphatase, warfarin therapy, frequent drops in blood pressure during hemodialysis, hypercoagulable conditions, corticosteroid and immunosuppressant therapy, calcium-phosphate product of more than 60 mg2/dL2, and use of blood products.5-8 Calcium-phosphate product is the most sensitive risk factor in the diagnosis of calciphylaxis, and small vessel calcification.5,9
BMI is an important predictor of calciphylaxis. Only 5% of hemodialysis patients have a BMI greater than 35 kg/m2 and calciphylaxis develops in 57% of these obese patients.6 Skin trauma, such as that caused by subcutaneous insulin injections in areas vulnerable to calciphylactic lesions, can also precipitate lesion development.6
Treatment. The current best therapy is prevention. Otherwise, treatment should be guided and modified subjectively. The high mortality and diagnostic difficulty will continue to challenge the physicians and dictate future research in this field.
References:
1. Fine A, Zacharias J. Calciphylaxis is usually non-ulcerating: risk factors, outcome and therapy. Kidney Int. 2002;61(6):2210-2217.
2. Selye H. Calciphylaxis. Chicago, IL: University of Chicago Press; 1962.
3. Budisavljevic MN, Cheek D, Ploth DW. Calciphylaxis in chronic renal failure. J Am Soc Nephrol. 1996;7(7):978-982.
4. Edelstein CL, Wickham MK, Kirby PA. Systemic calciphylaxis presenting as a painful, proximal myopathy. Postgrad Med J. 1992;
68(797):209-211.
5. Verdalles Guzmán U, de la Cueva P, Verde E, et al. [Calciphylaxis: fatal complication of cardiometabolic syndrome in patients with end stage kidney disease]. Nefrologia. 2008;28(1):32-36.
6. Parker RW, Mouton CP, Young DW, Espino DV. Early recognition and treatment of calciphylaxis. South Med J. 2003;96(1):53-55.
7. Bleyer AJ, Choi M, Igwemezie B, et al. A case control study of proximal calciphylaxis. Am J Kidney Dis. 1998;32(3):376-383.
8. Weenig RH, Sewell LD, Davis MD, et al. Calciphylaxis: natural history, risk factor analysis, and outcome. J Am Acad Dermatol. 2007;56(4):569-579.
9. Fischer AH, Morris DJ. Pathogensis of calciphylaxis: study of three cases with literature review. Hum Pathol. 1995;26(10):
1055-1064.


