Peer Reviewed
Breast Cancer Survivors: Physical, Psychological, Social, and Behavioral Care
AUTHORS:
Dawn M. Mussallem, DO; Julia A. Files, MD; and Sandhya Pruthi, MD
CITATION:
Mussallem DM, Files JA, Pruthi S. Breast cancer survivors: physical, psychological, social, and behavioral care. Consultant. 56(12):1086-1092.
ABSTRACT: To address the needs of the growing population of breast cancer survivors, the implementation of survivorship care guidelines for primary care providers that are clear, concise, and manageable is vital. Recently published breast cancer survivorship care guidelines can help primary care providers and breast cancer specialists coordinate care; they also delineate surveillance guidelines and management options for the adverse effects associated with cancer-related therapies and emphasize the importance of counseling and healthy lifestyle changes. The principal goals of the guidelines are to acknowledge cancer survivorship as a public health issue, to improve continuity of clinical care, and ultimately to help breast cancer survivors achieve optimal health and quality of life.
KEYWORDS: Breast cancer, cancer survivorship, cancer surveillance
Breast cancer survivorship is a dynamic experience that involves a patient’s journey from diagnosis onward and is experienced physically, psychologically, socially, behaviorally, and spiritually. To eliminate gaps in survivorship care, the American Cancer Society and the American Society of Clinical Oncology (ASCO) recently have published breast cancer survivorship care guidelines.1 These guidelines highlight the surveillance recommendations for recurrent and secondary cancers, the assessment of the long-term physical and psychosocial effects of cancer and cancer therapy, and the counseling of patients about preventive strategies. A survivorship care plan (SCP) will facilitate care coordination efforts, assist primary care teams, and support patients who have a history of early-stage breast cancer, with the goal of improving the quality of life of breast cancer survivors.
Epidemiology
Approximately 12.4% of women will receive a diagnosis of breast cancer at some point during their life.2 Early detection strategies and advances in breast cancer therapy have resulted in improved long-term survival rates and have increased the 5-year survival rate to almost 90%.2 Breast cancer survivors account for almost half of all female cancer patients, with an estimated 3.1 million breast cancer survivors currently in the United States.3,4
Cancer survivors are routinely part of a primary care provider’s patient population; however, nearly 47.9% of primary care physicians feel unprepared to manage the long-term effects of cancer treatment.5 Clinical practice guidelines are an essential means of guiding the primary care management of cancer survivors. Key components of these guidelines should highlight surveillance recommendations, outline the possible long-term effects of a patient’s past oncologic treatments, and emphasize preventive care and wellness.6-8
Coordination of Survivorship Care
The Institute of Medicine (IOM) report, From Cancer Patient to Cancer Survivor: Lost in Transition,9 states that “an individual is considered a cancer survivor from the time of cancer diagnosis through the balance of his or her life.”
Communication between the cancer specialist and the primary care provider and a clear understanding of both providers’ role and responsibilities are vital.10 SCPs are recommended in the IOM report for all cancer survivors and are designed to ensure care coordination and continuity of survivorship care. The goals of SCPs include an outline of the recommended surveillance/screening tests for the prevention and detection of recurrent cancers and secondary cancers and the assessment of the medical and psychosocial late effects of cancer therapy. Other goals of SCPs are to encourage patient adherence with long-term treatment and to emphasize optimal wellness. Although the intent of SCPs is to facilitate the coordination of care, enhance patient satisfaction, and communicate screening recommendations, there is a lack of evidence supporting their association with an improvement in overall care.11,12
The American College of Surgeons Commission on Cancer13 now mandates SCPs as a condition of accreditation of cancer facilities. Debate remains over what data should be included in SCPs, however. The Commission on Cancer’s Accreditation Committee determined that the ASCO data set (Table 1)13-15 represents the minimal content required by the standards, but this does not preclude programs from adding additional data elements.
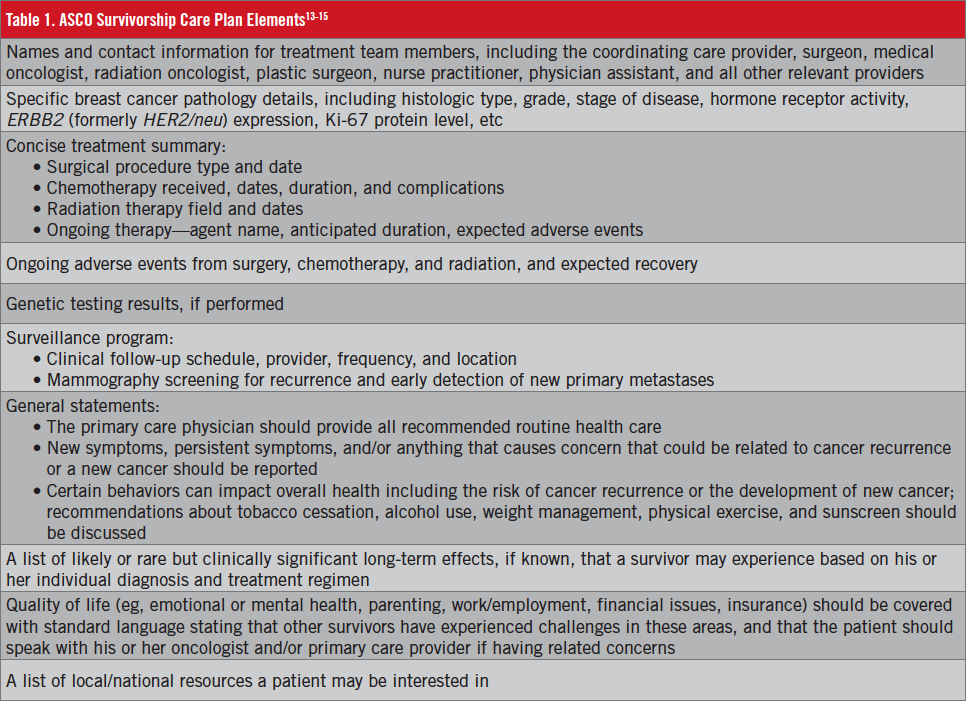
The SCP is completed by the provider or providers who coordinated the oncology treatment for the patient, with input from the patient’s other care providers. Some programs have established multidisciplinary survivorship clinics led by a nurse practitioner, physician assistant, or registered nurse. Although published guidelines do not endorse a single best practice, consensus appears to exist that the treating provider is central to the process. The SCP should be reviewed with the patient, and a copy sent to the primary care provider. Patients with metastatic disease are not delivered SCPs, given that their care remains with the oncologist.
Primary care providers or oncologists
Patients with early-stage breast cancer may follow up exclusively with a primary care provider. Care may be transferred approximately 1 year after the diagnosis. SCPs outline follow-up goals highlighting an optimal management strategy, enabling comprehensive transitioning of care. Periodic referral for oncologic assessment may be necessary for patients receiving adjuvant hormonal therapy to address questions regarding the safety of interventions, to address adverse events, and when suspicion or evidence of recurrent disease or secondary disease arises.
In studies conducted in the United Kingdom,16-18 the transfer of routine follow-up care to primary care physicians resulted in no increase in the time to diagnose a recurrent disease and no changes in patient anxiety level or other health-related quality of life domains. There was noted improvement in patient satisfaction and a reduction in patient and health-service costs. No data exist on outcomes of patients receiving primary care from nurse practitioners or physician assistants.
SURVIVORSHIP FOLLOW-UP
ASCO published its original breast cancer survivorship guidelines in 1997; these guidelines were updated in 1999, 2006, and 2012 (Table 2).19,20

History. The survivor’s age, specific cancer diagnosis, and treatment previously received should be specifically requested, and new symptoms that could represent local recurrence or metastatic disease should be investigated. Additionally, the psychosocial aspects of health should be targeted, including changes in mood, family, career, or living environment. A comprehensive review of systems should include the following:
- Constitutional symptoms such as weight fluctuations, fatigue, and changes in sleep patterns
- Cardiovascular symptoms such as chest pressure, edema, palpitations, and orthopnea
- Pulmonary symptoms such as chronic cough and shortness of breath
- Gastrointestinal tract symptoms such as right upper quadrant abdominal pain, nausea, vomiting, abdominal fullness, and changes in bowel patterns
- Bone health, including bone pain, arthralgias, and fracture
- Neurologic symptoms such as new headache, weakness, numbness, and tingling
- Gynecologic or genitourinary symptoms such as vaginal atrophy (dryness), dyspareunia, dysfunctional vaginal bleeding, pelvic pain, fertility concerns, sexual dysfunction, and changes in urinary patterns
- Psychological symptoms such as depression, anxiety, and distress
Physical examination. The clinical breast examination should be performed while the patient is upright, and then with the patient in the supine position. It should include examination of the affected breast, if preserved; in women who have undergone mastectomy, a thorough examination of the reconstructed breast is essential and must focus on the chest wall, the skin, and subcutaneous tissue, assessing the incision and ensuring that it is smooth. The examination also should include assessment of the contralateral breast, the chest wall, and the lymph nodes, including bilateral inspection of the axillary, cervical, supraclavicular, and infraclavicular regions.
The breast examination of women who have undergone adjuvant radiation therapy can be challenging. Diagrams of the affected breast can aid in following postoperative and postradiation changes over time. Women who have undergone mastectomy with or without breast reconstruction should be examined visually, and the incision site and surrounding skin of the chest wall should be palpated and inspected for abnormalities and signs of local recurrence, such as a new lump on the skin along the surgical scar or the nodal regions.
Breast Imaging
Mammography. Surveillance mammography is associated with a reduction in mortality among women of all ages.21 The purpose of posttreatment annual mammographic surveillance is to detect ipsilateral local recurrence after breast-conserving therapy and screen the contralateral unaffected breast. This should be performed annually.
Breast ultrasonography. Routine use of breast ultrasonography screening is not recommended. The American College of Radiology Imaging Network 6666 study22 compared the diagnostic yield of screening breast ultrasonography plus mammography vs mammography alone in women at elevated risk of breast cancer. The trial showed that the addition of breast ultrasonography screening to mammography will yield an additional 1.1 to 7.2 cancers per 1000 high-risk women, but it will also substantially increase the number of false positives from 4.4% to 10.4% and has a lower positive predictive value.
Breast magnetic resonance imaging (MRI). Breast MRI is not routinely recommended for breast cancer survivors, given the lack of evidence about its role in this population. A 2012 systematic review23 that included 10 case series enrolling a total of 494 patients revealed that the sensitivity and specificity of MRI for detecting recurrent breast cancer was no better than for mammography. Breast MRI is, however, indicated in women with an elevated risk of breast cancer based on family history models when the risk score exceeds 20%, in women with a known BRCA gene mutation, or in women whose original cancer was mammographically occult.8,23 Clinical breast examination remains the primary recommended means of detecting recurrent breast cancer after mastectomy with reconstruction. MRI and other diagnostic modalities should be used only to assess physical findings of concern.
The ASCO guidelines do not support the use of routine laboratory studies, tumor-marker evaluations, and radiologic imaging studies beyond mammography in asymptomatic patients. Such investigative tests have not been shown to increase survival, improve quality of life, or significantly shorten time to detection of recurrence. However, such testing may be necessary to evaluate specific symptoms on an individual basis.24
Since 2000, 5 randomized clinical trials involving 4023 women with early-stage breast cancer have compared routine follow-up with physical examination and mammography vs an intensive strategy, including radiologic and laboratory testing.25 No difference in overall survival was demonstrated between the groups (hazard ratio [HR], 0.96; 95% CI, 0.80-1.15) or disease-free survival (HR, 0.84; 95% CI, 0.71-1.00).
Bone Health
The ASCO guidelines highlight the importance of bone health optimization in breast cancer survivors with high-risk osteoporosis features, including age above 65 years; age between 60 to 64 years with a family history of osteoporosis; a body weight less than 70 kg; a prior history of nontraumatic fracture; and premenopausal women on tamoxifen and young cancer survivors with premature menopause.
Additionally, postmenopausal women with estrogen receptor–positive breast cancers are commonly treated with aromatase inhibitors (AIs), which are antiestrogen agents that reduce the systemic estrogen level by preventing its peripheral production. AI-associated bone loss occurs at more than twice the rate of postmenopause-related bone loss; therefore, vigilant management of bone health in patients who are being treated with AIs is essential.26
The ASCO guidelines recommend a baseline screening test for bone loss using dual energy x-ray absorptiometry in all high-risk patients as part of the annual survivorship evaluation, and then every 1 to 2 years depending on the specific individual and the specific osteoporosis risk factors present.27
Preventing Recurrence: Lifestyle Modification
Obesity, diet, and physical activity. Women who are obese at the time of diagnosis and women who gain weight after a breast cancer diagnosis are at a higher risk for breast cancer recurrence and death compared with women with a normal body mass index (BMI). Levels of estrogens, androgens, insulin-like growth factors, and insulin all increase with increased body fat and may promote breast cancer growth.28 ASCO urges oncologists and primary care providers to counsel overweight and obese women about diet and exercise.
Physical activity in survivors has been shown to be associated with a lower risk of breast cancer recurrence and death. Multiple studies have revealed that exercise improves the quality of life of breast cancer survivors.7,29 Therefore, patients should be encouraged to adopt a physically active lifestyle following treatment for breast cancer. Exercise regimens should include aerobic and resistance training. Any activity that gets a person moving, such as bicycling, walking, mowing the lawn, or sports participation is acceptable. The US Department of Health and Human Services Physical Activity Guidelines for Americans, for example, recommend 150 minutes of moderate-intensity aerobic exercise per week and 2 weekly sessions of weight training for the general population.30,31
The importance of eating healthy whole foods should be emphasized. Dietary recommendations should focus on a diet rich in fruits and vegetables, whole grains, low-fat dairy, nuts, beans, wild fish, lean meat, and minimization of red meat.32 Patients with a BMI greater than 30 kg/m2 should be offered dietitian consultation and a comprehensive supervised lifestyle intervention program that includes a reduced-calorie diet of 1200 to 1500 calories, increased physical activity, and behavioral strategies. If medically supervised comprehensive lifestyle interventions are not available to the patient, electronically delivered interventions that provide personalized feedback by a trained interventionist, or a commercial program using counseling, may be considered, after ensuring that these interventions have outcomes supported by scientific evidence of safety and efficacy.
Stress management. Psychosocial distress in cancer survivors is common. All patients should have mental health screening (eg, Generalized Anxiety Disorder 7-item scale, Patient Health Questionnaire) at regular intervals. If symptoms of depression or anxiety are present, further investigation is required to determine the source of the symptoms and any potential interventions. Resources such as stress-reduction classes; consultation with social workers, chaplains, psychologists, or psychiatrists; and support groups (online or in person) should be offered to survivors.33
Alcohol consumption. Alcohol is thought to be associated with an increase in the risk of breast cancer recurrence.15 In the Life After Cancer Epidemiology (LACE) study,34 drinking the equivalent of 3 to 4 standard alcoholic drinks or more per week was associated with a 1.3-fold increased risk of breast cancer recurrence and 1.5-fold increased risk of breast cancer death. Overweight and postmenopausal women seemed to experience the greatest harm from alcohol intake as measured by breast cancer recurrence risk.
Menopausal symptoms
Vasomotor symptoms and vaginal dryness may occur as a result of chemotherapy-induced premature menopause. They may also occur in premenopausal and postmenopausal women on antiestrogen therapy.
Although it is not approved by the US Food and Drug Administration (FDA) for the treatment of hot flashes, venlafaxine is considered a first-line nonhormonal medication.35-37 Initiation of selective serotonin-reuptake inhibitors (SSRIs) for hot flash management should be avoided in women who are taking tamoxifen, because these medications can interfere with cytochrome P450 2D6 (CYP2D6) metabolism of tamoxifen to its active form.38 Women with depression who are on tamoxifen and an SSRI should be transitioned to an alternative antidepressant or an SSRI with minimal inhibitory CYP2D6 activity, such as citalopram. Abrupt cessation of SSRIs should be avoided and guided by a health care professional.39 Another acceptable treatment option is gabapentin.40 The adverse effects associated with gabapentin include lightheadedness. Nonpharmaceutical options include dressing in layers, using a fan or air conditioner to keep air flowing, daily exercise, and avoiding consumption of spicy foods, caffeine, and alcohol. Smoking is also linked with an increased incidence of hot flashes.
Patients may also be referred to a specialist in complementary and alternative medicine to manage hot flashes. Acupuncture can be an effective integrative intervention for managing hot flashes and improving the quality of life in women with breast cancer.41
Vaginal Atrophy, Dyspareunia, Sexual Dysfunction
Vaginal moisturizers must be used every few days for maintenance of vaginal tissue integrity. Hyaluronic acid vaginal gel has been shown to be noninferior to vaginal estrogen cream with respect to improvement in vaginal dryness, dyspareunia, and burning. Hyaluronic acid vaginal gel may be considered as an alternative to estrogen-based treatments for relieving symptoms of vaginal dryness.42
Over-the-counter vaginal lubricants can be applied to the vaginal introitus and to the partner at the time of sexual activity to decrease friction or vaginal pain. Repeat application during sexual activity may be required. Avoid lubricants with warming sensations, flavors, and known irritants such as glycerin, parabens, and propylene glycol.
A randomized study evaluating the use of dehydroepiandrosterone (DHEA) for the management of vaginal symptoms in cancer survivors reported an improvement in vaginal atrophy symptoms and sexuality compared with the use of a vaginal moisturizer alone.43 The therapy was well tolerated, but mild hirsutism and increased acne were attributed to the use of DHEA. Circulating estradiol in patients receiving AIs did not substantially increase with DHEA use; however, these levels did increase slightly in women not on AI therapy.43
Whether vaginal estrogen increases the risk of breast cancer recurrence and whether it is safe for long-term use remain unknown. Vaginal estrogen in general should be avoided in women on AI therapy. If all other nonestrogen options for vaginal dryness have been exhausted and the patient remains symptomatic, discontinuation of AI therapy should be considered, and tamoxifen therapy should be considered if appropriate. If vaginal dryness persists and impacts the patient’s quality of life, low-dose vaginal estrogen can be prescribed with the cautions noted above. In this scenario, a rise in circulating estradiol would be more acceptable given that tamoxifen is an estrogen-receptor antagonist with partial agonist activity.44
Hormonal options for symptomatic patients include vaginal estradiol tablets, 10 µg, once or twice weekly, or an estradiol vaginal ring inserted for 3 continuous months. Applying a pearl-sized amount of estrogen cream to the vaginal introitus 2 or 3 times weekly may be sufficient to relieve dyspareunia symptoms; however, vaginal insertion of the cream should be avoided.
Topical lidocaine ointment or gel may be used in combination with other therapies at the introitus to relieve insertional pain, preferably in conjunction with other treatment modalities such as moisturizers, lubricants, and pelvic floor physical therapy. Women should be cautioned not to cover up pain when using lidocaine gel.
Referral to a sexual health and/or mental health specialist may also be helpful.
Fertility and Contraception
Tamoxifen is an FDA-approved drug in pregnancy category D. Although no controlled data exist on human pregnancy, animal studies have revealed evidence of fetotoxicity, a lower incidence of implantation, and an increased incidence of low birth weight. Women of reproductive age who are prescribed tamoxifen should receive counseling about pregnancy avoidance and effective contraception that is compatible with tamoxifen use.45 These contraceptive options include copper intrauterine devices (preferred), sterilization (male or female), and a double-barrier method (condom plus cervical cap or diaphragm). Combined hormonal contraception should be avoided because of the risk of venous thromboembolism (VTE) associated with tamoxifen. Oral progestin-only contraceptives may lead to an increased risk of VTE, but this is not certain. Injectable progestin appears to lead to an increased risk of VTE.
AIs are FDA-approved drugs in pregnancy category C. Animal reproduction studies have shown an adverse effect on the fetus, but no adequate or well-controlled studies have been performed in humans. Any woman of childbearing potential who is taking AIs should be on concurrent ovarian function-suppressing medications, but this is only effective for contraception if ovarian function is completely suppressed.
Patients who have a history of estrogen receptor–negative breast cancer and bilateral mastectomies may opt to take hormonal contraception, but oral hormonal contraception is discouraged for all other breast cancer survivors.
Levonorgestrel-releasing intrauterine systems are known to slightly increase systemic hormone levels and thus are discouraged for patients with a history of hormone-sensitive breast cancer unless they are receiving tamoxifen.46-48 Levonorgestrel is one of the most potent progestins available, and it is measureable in the serum of women with these devices. In women whose intrauterine system has become dislodged into the peritoneal cavity, serum levonorgestrel levels can be very high. Clinicians should discuss the use of a nonhormonal contraceptive, including barrier protection and copper intrauterine devices, to help a woman choose the method most consistent with her lifestyle and beliefs.
Genetic Counseling
A detailed oncologic family history should be collected and updated periodically.
Referral to a genetic counselor for further genetic risk assessment, as well as consideration of genetic testing, should be considered for all women receiving a diagnosis of breast cancer before age 50, for women with triple-negative breast cancer diagnosed at age 60 or before, for women with a history of 2 primary breast cancers, and for women with a known mutation of a breast cancer–susceptibility gene in their family history.
Additionally, genetic counseling referral should be sought for women with a diagnosis of breast cancer at any age and who have at least 1 first-, second-, or third-degree relative with breast cancer before age 50 or invasive ovarian cancer at any age; for women with at least 2 first-, second-, or third-degree relatives with breast cancer or pancreatic cancer at any age; for women from a population at increased risk; and for women of Ashkenazi Jewish descent with breast, ovarian, or pancreatic cancer at any age.49
Dawn M. Mussallem, DO, is a consultant in the Department of Hematology and Oncology and the Robert and Monica Jacoby Center for Breast Health and an assistant professor of medicine at the Mayo Clinic in Jacksonville, Florida.
Julia A. Files, MD, is a consultant in the Medallion program, an associate professor of medicine, and former chair of the Division of Women’s Health in the Department of Internal Medicine at the Mayo Clinic in Scottsdale, Arizona.
Sandhya Pruthi, MD, is a consultant in the Division of General Internal Medicine, former medical director of patient experience, and a professor of medicine at the Mayo Clinic in Rochester, Minnesota.
References:
- Runowicz CD, Leach CR, Henry NL, et al. American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline. J Clin Oncol. 2016;34(6):611-635.
- SEER stat fact sheets: female breast cancer. National Institutes of Health; National Cancer Institute; Surveillance, Epidemiology, and End Results Program. http://seer.cancer.gov/statfacts/html/breast.html. Accessed November 1, 2016.
- Ganz PA. Survivorship: adult cancer survivors. Prim Care. 2009;36(4):721-741.
- Miller KD, Siegel RL, Lin CC, et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016;66(4):271-289.
- Bober SL, Recklitis CJ, Campbell EG, et al. Caring for cancer survivors: a survey of primary care physicians. Cancer. 2009;115(18 suppl):4409-4418.
- Moyer VA; US Preventive Services Task Force. Risk Assessment, genetic counseling, and genetic testing for BRCA-related cancer in women: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(4):271-281.
- Rock CL, Doyle C, Demark-Wahnefried W, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62(4):243-274.
- Saslow D, Boetes C, Burke W, et al; American Cancer Society Breast Cancer Advisory Group. American Cancer Society Guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57(2):75-89.
- Hewitt M, Greenfield S, Stovall E, eds; Committee on Cancer Survivorship: Improving Care and Quality of Life; National Cancer Policy Board. From Cancer Patient to Cancer Survivor: Lost in Transition. Washington, DC: National Academies Press; 2006:19.
- Cheung WY, Neville BA, Cameron DB, Cook EF, Earle CC. Comparisons of patient and physician expectations for cancer survivorship care. J Clin Oncol. 2009;27(15):2489-2495.
- Brennan ME, Gormally JF, Butow P, Boyle FM, Spillane AJ. Survivorship care plans in cancer: a systematic review of care plan outcomes. Br J Cancer. 2014;111(10):1899-1908.
- Bulloch KJ, Irwin ML, Chagpar AB, et al. Systematic approach to providing breast cancer survivors with survivorship care plans: a feasibility study. J Oncol Pract. 2015;11(2):e170-e176.
- American College of Surgeons Commission on Cancer. Cancer Program Standards: Ensuring Patient-Centered Care. Chicago, IL: American College of Surgeons; 2016.
- American College of Surgeons. Accreditation Committee clarifications for standard 3.3 survivorship care plan. https://www.facs.org/publications/newsletters/coc-source/special-source/standard33. Published September 9, 2014. Accessed October 31, 2016.
- Scoccianti C, Lauby-Secretan B, Bello PY, Chajes V, Romieu I. Female breast cancer and alcohol consumption: a review of the literature. Am J Prev Med. 2014;46(3 suppl 1):S16-S25.
- Grunfeld E, Gray A, Mant D, et al. Follow-up of breast cancer in primary care vs specialist care: results of an economic evaluation. Br J Cancer. 1999;79(7-8):1227-1233.
- Grunfeld E, Mant D, Yudkin P, et al. Routine follow up of breast cancer in primary care: randomised trial. BMJ. 1996;313(7058):665-669.
- Kerrigan D, Waters P, Ryan M, et al. Follow-up arrangements for breast cancer patients; is it appropriate to transfer surveillance to general practitioners? Ir Med J. 2014;107(9):273-275.
- Khatcheressian JL, Hurley P, Bantug E, et al. Breast cancer follow-up and management after primary treatment: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(7):961-965.
- Runowicz CD, Leach CR, Henry NL, et al. American Cancer Society/American Society of Clinical Oncology breast cancer survivorship care guideline. J Clin Oncol. 2016;34(6):611-635.
- Lash TL, Fox MP, Silliman RA. Reduced mortality rate associated with annual mammograms after breast cancer therapy. Breast J. 2006;12(1):2-6.
- Berg WA, Blume JD, Cormack JB, et al; ACRIN 6666 Investigators. Combined screening with ultrasound and mammography vs mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299(18):2151-2163.
- Quinn EM, Coveney AP, Redmond HP. Use of magnetic resonance imaging in detection of breast cancer recurrence: a systematic review. Ann Surg Oncol. 2012;19(9):3035-3041.
- Gradishar WJ, Anderson BO, Balassanian R, et al. Invasive Breast Cancer Version 1.2016, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2016;14(3):324-354.
- Moschetti I, Cinquini M, Lambertini M, Levaggi A, Liberati A. Follow-up strategies for women treated for early breast cancer. Cochrane Database Syst Rev. 2016(5):CD001768. doi:10.1002/14651858.CD001768.pub3.
- Hadji P. Aromatase inhibitor-associated bone loss in breast cancer patients is distinct from postmenopausal osteoporosis. Crit Rev Oncol Hematol. 2009;69(1):73-82.
- Pant S, Shapiro CL. Aromatase inhibitor-associated bone loss: clinical considerations. Drugs. 2008;68(18):2591-2600.
- Caan B, Sternfeld B, Gunderson E, Coates A, Quesenberry C, Slattery ML. Life After Cancer Epidemiology (LACE) Study: a cohort of early stage breast cancer survivors (United States). Cancer Causes Control. 2005;16(5):545-556.
- Sternfeld B, Weltzien E, Quesenberry CP Jr, et al. Physical activity and risk of recurrence and mortality in breast cancer survivors: findings from the LACE study. Cancer Epidemiol Biomarkers Prev. 2009;18(1):87-95.
- Arem H, Moore SC, Patel A, et al. Leisure time physical activity and mortality: a detailed pooled analysis of the dose-response relationship. JAMA Intern Med. 2015;175(6):959-967.
- Schmid D, Leitzmann MF. Association between physical activity and mortality among breast cancer and colorectal cancer survivors: a systematic review and meta-analysis. Ann Oncol. 2014;25(7):1293-1311.
- Pierce JP, Natarajan L, Caan BJ, et al. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: the Women’s Healthy Eating and Living (WHEL) randomized trial. JAMA. 2007;298(3):289-298.
- Clark EJ, Stovall EL. Advocacy: the cornerstone of cancer survivorship. Cancer Pract. 1996;4(5):239-244.
- Kwan ML, Kushi LH, Weltzien E, et al. Alcohol consumption and breast cancer recurrence and survival among women with early-stage breast cancer: the Life After Cancer Epidemiology study. J Clin Oncol. 2010;28(29):4410-4416.
- Biglia N, Torta R, Roagna R, et al. Evaluation of low-dose venlafaxine hydrochloride for the therapy of hot flushes in breast cancer survivors. Maturitas. 2005;52(1):78-85.
- Loprinzi CL, Kugler JW, Sloan JA, et al. Venlafaxine in management of hot flashes in survivors of breast cancer: a randomised controlled trial. Lancet. 2000;356(9247):2059-2063.
- Ramaswami R, Villarreal MD, Pitta DM, Carpenter JS, Stebbing J, Kalesan B. Venlafaxine in management of hot flashes in women with breast cancer: a systematic review and meta-analysis. Breast Cancer Res Treat. 2015;152(2):231-237.
- Henry NL, Stearns V, Flockhart DA, Hayes DF, Riba M. Drug interactions and pharmacogenomics in the treatment of breast cancer and depression. Am J Psychiatry. 2008;165(10):1251-1255.
- Kelly CM, Juurlink DN, Gomes T, et al. Selective serotonin reuptake inhibitors and breast cancer mortality in women receiving tamoxifen: a population based cohort study. BMJ. 2010;340:c693.
- Johns C, Seav SM, Dominick SA, et al. Informing hot flash treatment decisions for breast cancer survivors: a systematic review of randomized trials comparing active interventions. Breast Cancer Res Treat. 2016;156(3):415-426.
- Lesi G, Razzini G, Musti MA, et al. Acupuncture as an integrative approach for the treatment of hot flashes in women with breast cancer: a prospective multicenter randomized controlled trial (AcCliMaT). J Clin Oncol. 2016;34(15):1795-1802.
- Chen J, Geng L, Song X, Li H, Giordan N, Liao Q. Evaluation of the efficacy and safety of hyaluronic acid vaginal gel to ease vaginal dryness: a multicenter, randomized, controlled, open-label, parallel-group, clinical trial. J Sex Med. 2013;10(6):1575-1584.
- Barton DL, Sloan JA, Shuster LT, et al. Impact of vaginal dehydroepiandosterone (DHEA) on vaginal symptoms in female cancer survivors: Trial N10C1 (Alliance). Presented at: Annual Meeting of the American Society of Clinical Oncology; May 30, 2014; Chicago, IL. Abstract 9507. http://meetinglibrary.asco.org/content/125315-144. Accessed November 1, 2016.
- American College of Obstetricians and Gynecologists Committee on Gynecologic Practice. Committee opinion No. 659: the use of vaginal estrogen in women with a history of estrogen-dependent breast cancer. Obstet Gynecol. 2016;127(3):e93-e96.
- Braems G, Denys H, De Wever O, Cocquyt V, Van den Broecke R. Use of tamoxifen before and during pregnancy. Oncologist. 2011;16(11):1547-1551.
- Schwarz EB, Hess R, Trussell J. Contraception for cancer survivors. J Gen Intern Med. 2009;24(suppl 2):S401-S406.
- Blamey RW, Jonat W, Kaufmann M, Bianco AR, Namer M. Goserelin depot in the treatment of premenopausal advanced breast cancer. Eur J Cancer. 1992;28A(4-5):810-814.
- Carlson RW, Theriault R, Schurman CM, et al. Phase II trial of anastrozole plus goserelin in the treatment of hormone receptor–positive, metastatic carcinoma of the breast in premenopausal women. J Clin Oncol. 2010;28(25):3917-3921.
- Daly MB, Pilarski R, Axilbund JE, et al. Genetic/familial high-risk assessment: breast and ovarian, version 2.2015. J Natl Compr Canc Netw. 2016;14(2):153-162.


