Are These Lumps Benign or Reason for Worry?
Photo Essay
Cervical Lymphadenitis
A 4-year-old girl presented with a low-grade fever, a sore throat, and a painful mass in the left cervical area for a few days. There was no history of cat scratch. She was otherwise in good health and was not on any medication. She did not have lymph node enlargement elsewhere. On examination, the cervical mass was very tender and not fixed to the skin or underlying structure. Her throat was beefy red, and exudates were visible on the tonsils. Her tongue was strawberry-like. Physical examination was otherwise normal. In particular, there was no hepatosplenomegaly.

The child had cervical lymphadenitis. Acute bilateral cervical lymphadenitis usually is caused by a viral upper respiratory tract infection or pharyngitis due to Streptococcus pyogenes. Acute unilateral cervical lymphadenitis usually is caused by S pyogenes or Staphylococcus aureus.1 A beefy red tongue, tonsillar exudates, petechiae on the hard palate, and a strawberry tongue suggest infection caused by S pyogenes.2 The patient was treated with amoxicillin 250 mg three times a day for 10 days, which led to resolution of the cervical lymphadenitis.
Because of its proven efficacy, safety, and narrow spectrum of antimicrobial activity, penicillin V remains the drug of choice for cervical lymphadenitis caused by S pyogenes. Amoxicillin often is preferred over penicillin V because of the suspension’s better taste and its availability as chewable tablets.2
Treatment of acute bacterial cervical lymphadenitis without a known primary infectious source should provide adequate coverage for both S aureus and S pyogenes, pending results of culture and sensitivity tests if available.3 Appropriate oral antibiotics include cloxacillin, cephalexin, cefprozil, or clindamycin.3 Children with cervical lymphadenopathy and periodontal or dental disease should be treated with clindamycin or a combination of amoxicillin and clavulanic acid, which provide coverage for anaerobic oral flora.3
REFERENCES:
1. Leung AK, Davies HD. Cervical lymphadenitis: etiology, diagnosis, and management. Curr Infect Dis Rep. 2009;11(3):183-189.
2. Leung AK, Kellner JD. Group A beta-hemolytic streptococcal pharyngitis in children. Adv Ther. 2004;21(5):277-287.
3. Peters TR, Edwards KM. Cervical lymphadenopathy and adenitis. Pediatr Rev. 2000:21(12):399-405.
Rhabdomyosarcoma
A 7-year-old boy presented with right-sided facial swelling, epistaxis, and purulent discharge from the right nostril. There was no history of fever, night sweating, or weight loss. He had no exposure to individuals with an infectious illness.
On examination, the child was afebrile. The right side of his face was swollen, slightly warm, and mildly tender. The right nostril was full of purulent material. The right maxillary molars were very mobile, and the adjacent gingival tissue was swollen. The soft palate on the right side was displaced downward.
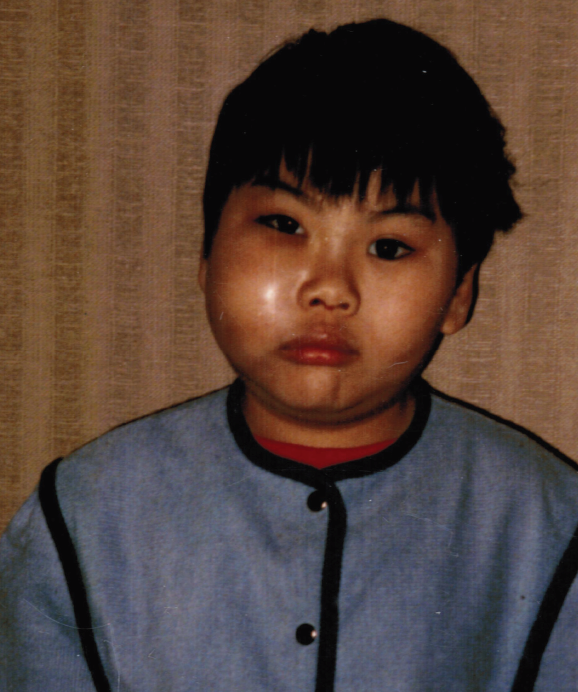
A radiograph of the sinuses showed opacification of the right maxillary sinus, complete loss of the cortical margin of the right maxillary antrum, elevation of the right orbital floor, and a soft tissue mass in the right naris with destruction of the right nasal arch in its inferior portion. Antrostomy and biopsy of the mass were done. Pathologic examination revealed a rhabdomyosarcoma.
Rhabdomyosarcoma is a malignant tumor of the same embryonic mesenchyme that gives rise to striated skeletal muscle.1,2 It is the most common soft tissue sarcoma in children and adolescents, and it accounts for 3.5% of cancers in children up to 14 years of age and 2% in adolescents up to 19 years of age.1,2 Rhabdomyosarcoma affects 250 to 300 children per year in the United States.3 Approximately 75% of cases occur in children under age 12, and 50% occur in children under age 5.4 The head and neck region is the most frequent site, accounting for approximately 40% of cases.4,5
The 3 main histologic types of rhabdomyosarcoma are embryonal, alveolar, and pleomorphic.4,5 Embryonal rhabdomyosarcoma commonly affects younger children, and alveolar rhabdomyosarcoma is more frequent in adolescents. Pleomorphic rhabdomyosarcoma occurs mainly in adults.4 Embryonal rhabdomyosarcoma develops primarily in the head and neck area, whereas the alveolar and the pleomorphic varieties are more common in the extremities.4
Because of the characteristic rapid growth of the tumor, purulent nasal discharge, and its occasional association with pain, rhabdomyosarcoma arising from the maxillary sinus may be mistaken for maxillary sinusitis secondary to a dental abscess.1 Other clinical features can include proptosis, diplopia, epistaxis, nasal obstruction, recurrent otitis media, and a unilateral polypoid mass in the maxillary antrum.2 Imaging studies such as computed tomography or magnetic resonance imaging are necessary to reveal the location and extent of the tumor.5
Treatment consists of a combination of chemotherapy, radiotherapy, and surgery.2 Surgery is a consideration in patients with resectable tumors or as salvage therapy in patients who do not respond completely to chemotherapy and radiation.2 The overall survival rate for rhabdomyosarcoma is approximately 60% at 5 years.4
REFERENCES:
1. Leung AKC. Rhabdomyosarcoma simulating dental abscess. Oral Surg Oral Med Oral Pathol. 1988;65(1):114.
2. Fyrmpas G, Wurm J, Athanassiadou F, et al. Management of paediatric sinonasal rhabdomyosarcoma. J Laryngol Otol. 2009;123(9):990-996.
3. Hayes-Jordan A, Andrassy R. Rhabdomyosarcoma in children. Curr Opin Pediatr. 2009;21(3):373-378.
4. Sinha R, Bandyopadhyay SN, Pathak K, Hati G. Rhabdomyosarcoma of nose and paranasal sinuses—a therapeutic challenge. J Indian Med Assoc. 2006;104(9):519-521.
5. Ahmed AA, Tsokos M. Sinonasal rhabdomyosarcoma in children and young adults. Int J Surg Pathol. 2007;15(2):160-165.
Sacrococcygeal Teratoma
An infant boy was born to a gravida 2, para 1, 23-year-old woman at 35 weeks of gestation. The mother was in good health and was not on any medication during the pregnancy. There was no history of consanguinity. Elective cesarean section was performed, because prenatal ultrasonography showed a large mass extending from the lower back of the fetus.
On examination, a huge mass was found extending from the coccyx of the infant, just posterior to the genitalia.
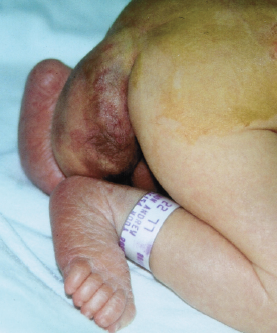 A sacrococcygeal teratoma usually presents as a large midline mass protruding between the coccyx and the anus, and it usually is covered with normal, intact skin.1 Rarely, the skin is necrotic owing to a compromise in blood supply.2 The anus usually is displaced anteriorly. Characteristically, the tumor comprises both solid and cystic elements, with more than 50% of tumors containing calcification and ossification.3 Other characteristics of sacrococcygeal teratoma include hemorrhage, necrosis, and cystic degeneration.3
A sacrococcygeal teratoma usually presents as a large midline mass protruding between the coccyx and the anus, and it usually is covered with normal, intact skin.1 Rarely, the skin is necrotic owing to a compromise in blood supply.2 The anus usually is displaced anteriorly. Characteristically, the tumor comprises both solid and cystic elements, with more than 50% of tumors containing calcification and ossification.3 Other characteristics of sacrococcygeal teratoma include hemorrhage, necrosis, and cystic degeneration.3
Sacrococcygeal teratomas can be classified into 4 distinct anatomic types that differ in the degree of intrapelvic and extrapelvic extension: type I, predominantly external with minimal presacral extension; type II, external but with significant intrapelvic extension; type III, primarily pelvic and abdominal, but apparent externally; and type IV, presacral with no external presentation.1
Approximately 2% to 10% of sacrococcygeal teratomas are malignant before the infant is 2 months of age, 50% are malignant by 1 year of age, and almost 100% are malignant by 5 years of age.1 Malignant transformation most likely occurs between 4 months and 5 years of age.4 Malignant change is more frequent in boys. A tumor with a large presacral component may cause gastrointestinal or urinary tract obstruction. Other complications can include anemia and high-output cardiac failure.3,5
Sacrococcygeal teratoma is the most common tumor in the neonatal period. The incidence is approximately 1 per 35,000 to 40,000 live births per year.4 The female to male ratio is approximately 4:1.1,2 Most cases occur sporadically, although 14% to 50% of patients have a family history of twinning.1,2
Teratomas are congenital tumors composed of tissues derived from the three primitive germinal layers: ectoderm, mesoderm, and endoderm. Postsacral tumors are thought to arise from the pluripotent embryonic cells that occur in greatest concentration in the Hensen node of the primitive knot, and presacral tumors from incomplete migration of germ cells from the yolk sac to the urogenital ridge.1-3 The female gonads terminate their differentiation at 10 weeks, in contrast to 7 weeks for the male testes. Their totipotential cells, therefore, remain longer and are more prone to disturbances. This might account for the higher incidence of sacrococcygeal teratomas in girls.2 Their midline distribution can be explained by the arrest or aberrant migration of the primordial germ cells.6 Sacrococcygeal teratomas may be the result of twinning or abortive attempts at twinning, which might account for the higher incidence in monozygotic twins.
The diagnosis usually is made during prenatal ultrasonography.3 Postnatally, the diagnosis is mainly clinical. Lateral abdominal radiograph may show anterior displacement of the rectum, sacral defects, and possible calcification within the tumor. Abdominal and pelvic ultrasonography, computed tomography, and magnetic resonance imaging help assess for any internal extension of the tumor, involvement of other organs, and the existence of metastatic lesions. Magnetic resonance imaging is particularly useful in the evaluation of spinal canal infiltration. A chest radiograph or computed tomography scan is useful for revealing pulmonary metastasis. Serum α-fetoprotein levels are elevated in 70% of children with malignant tumors, with normal levels in children with benign tumors.1
If the tumor is greater than 5 cm on prenatal ultrasonography, cesarean section is recommended to avoid dystocia and rupture of the tumor.1,3,5 Complete resection of the tumor and the coccyx is essential, because failure to remove the coccyx will result in local recurrence in 30% of cases.1 The most common complication after surgery is fecal incontinence. If the tumor is not resectable, chemotherapy may shrink the tumor and render it resectable. Adjunct chemotherapy also is indicated for malignant tumors at stage II or greater and for the treatment of distant metastasis.1 n
REFERENCES:
1. Leung AKC, Kao CP. Picture of the month: sacrococcygeal teratoma. Arch Pediatr Adolesc Med. 2000;154(3):309-310.
2. Leung AKC, Kao CP, Wong AL. Sacrococcygeal teratoma. In: Lang F, ed. Encyclopedia of Molecular Mechanisms of Disease. Berlin, Germany: Springer-Verlag; 2009:1879-1880.
3. Wee WW, Tagore S, Tan JVK, Yeo GSH. Foetal sacrococcygeal teratoma: extremes in clinical presentation. Singapore Med J. 2011;52(6):e118-e123.
4. Abubakar AM, Nggada HA, Chinda JY. Sacrococcygeal teratoma in Northeastern Nigeria: 18-years experience. Pediatr Surg Int. 2005;21(8):645-648.
5. Gucciardo L, Uyttebroek A, De Wever I, et al. Prenatal assessment and management of sacrococcygeal teratoma. Prenat Diagn. 2011;31(7):678-688.
6. Makin EC, Hyett J, Ade-Ajayi N, Patel S, Nicolaides K, Davenport M. Outcome of antenatally diagnosed sacrococcygeal teratomas: single-center experience (1993-2004). J Pediatr Surg. 2006;41(2):388-393.
Umbilical Granuloma
A 3-month-old infant presented with a soft, moist, velvety, dull red mass in the umbilical area. He was born to a gravida 2, para 1, 26-year-old mother following an uncomplicated pregnancy with full-term normal spontaneous delivery. The umbilical cord separated at 16 days of age. The neonatal course was uneventful.

The infant had an umbilical granuloma. An umbilical granuloma forms from excess granulation tissue persisting at the base of the umbilical area after separation of the umbilical cord. The condition represents continuing inflammation of the granulation tissue that has not yet epithelialized. Delayed separation of the umbilical cord increases the risk of granuloma formation. The presence of saprophytic organisms in the umbilical area increases the chance of invasion by pathogenic organisms.
Typically, an umbilical granuloma presents as a red papule or a pedunculated mass with a soft, velvety appearance. The lesion usually is from 3 to 10 mm in size. An umbilical granuloma must be differentiated from an umbilical polyp, which results from persistence of all or part of the omphalomesenteric duct.
The most common treatment is topical application of silver nitrate solution or 75% silver nitrate/25% potassium nitrate stick. Silver nitrate acts as an antiseptic, astringent, and caustic agent.1 Application of silver nitrate to the granuloma triggers cauterization.1 The procedure is simple and can be done easily in a physician’s office. Because of the possibility of chemical burns to the periumbilical area, caution should be exercised during silver nitrate application.
Ligation of the stalk just distal to its origin is another treatment option. However, most umbilical granulomas are deeply situated, which may render ligation of the granulomas difficult. A double-ligature technique can overcome the technical difficulty.2 The first stage involves placing a superficial “stay” ligature, which brings the umbilical granuloma into a more easily accessible position. The second stage involves placing a deeper and more precise ligature at the base of the lesion.
Other treatment options include alcoholic wipes at each diaper change, topical antibiotic ointment, cryosurgery, electrocautery, excision and application of absorbable hemostatic material, and salt crystals.
REFERENCES:
1. Nagar H. Umbilical granuloma: a new approach to an old problem. Pediatr Surg Int. 2001;17(7):513-514.
2. Lotan G, Klin B, Efrati Y. Double-ligature: a treatment for pedunculated umbilical granulomas in children. Am Fam Physician. 2002;65(10):2067-2068.


