American Academy of Pain Medicine (AAPM) 28th Annual Meeting
February 23-26, 2012; Palm Springs, CA
_____________________________________
Geriatric Patients Metabolize Oxymorphone Extended-Release Tablets More Slowly
A recent study provides evidence that older adults metabolize oxymorphone extended-release (ER) tablets at a much slower rate than younger adults, resulting in significantly higher and possibly dangerous mean and maximum plasma concentrations of the opioid’s two metabolites. Investigators, who presented data from the open-label trial during a poster session at the AAPM 28th Annual Meeting, also said that women metabolize oxymorphone ER more slowly than men of the same age. These findings might help explain why women appear more susceptible to the analgesic and gastrointestinal effects of opioid agents like oxymorphone.
To compare the effects of oxymorphone ER between younger and older adults and between men and women, the investigators enrolled 48 healthy individuals whose weight was within 15% of Metropolitan Life Insurance Company standards. All participants were nonsmokers with normal echocardiograms and laboratory results and no history of alcohol abuse, illicit drug use, or mental illness. The study included an equal number of men (n=24) and women (n=24). Half the men (n=12) and half the women (n=12) were between the ages of 20 and 40 years, and the remaining men (n=12) and women (n=12) ranged in age from 65 to 81 years.
On the first day of the study, patients received a 20-mg tablet of oxymorphone ER. Plasma and urine specimens were obtained at regular intervals over the next 48 hours to establish a single-dose pharmacokinetic profile of the drug. The second 20-mg dose of oxymorphone ER was administered on the morning of day 3, with subsequent doses given at 12-hour intervals until patients had taken 13 pills. During this phase of the study, participants also took naltrexone 50 mg each night to minimize oxymorphone’s opioid effects. Investigators continued to gather plasma and urine samples at specified intervals throughout the study and for 12 hours after participants took their last dose of oxymorphone.
In the study’s single-dose phase, older adults achieved a higher mean area under the curve (AUC) and maximum concentration (Cmax) of oxymorphone and of its two metabolites, 6-OH-oxymorphone (6-OH-Oxy) and oxymorphone-3-glucuronide (Oxy-3-G). Stratifying results by sex revealed a higher mean AUC and Cmax for oxymorphone and its metabolites in women than in men, but the difference between the sexes was not as significant as the difference between the age groups. The time to maximum concentration (tmax) was shorter in younger adults, and there was also a longer terminal elimination half-life in these individuals than in older adults.
Pharmacokinetic profiles in the steady-state phase of the study were similar: the mean steady-state AUC and Cmax for oxymorphone, 6-OH-Oxy, and Oxy-3-G were higher in older participants than in younger ones; and the mean AUC and Cmax of oxymorphone and Oxy-3-G were higher in women than in men. Overall, the steady-state findings showed mean and maximum plasma concentrations of oxymorphone and its metabolites were 35% to 40% higher for older adults versus younger adults, and women had mean plasma concentrations averaging 14% to 21% higher than concentrations observed in men. Older women had an especially long tmax for oxymorphone, at 5.0 hours versus 3.5 hours in older men, 2.5 hours in younger men, and 1.8 hours in younger women. The researchers assessed whether body weight might have affected the pharmacokinetics and determined that although it accounted for the sex-based differences in pharmacokinetics, it did not explain the age-based differences.
The authors note that guidelines from the American Geriatrics Society do not suggest specific doses when initiating older patients on opioid analgesics and instead recommend a trial introduction with careful monitoring due to the heterogeneity of older adults. Based on this study’s findings, the authors said, “Initiating oxymorphone ER at a lower dose in older patients is advisable to minimize adverse events.”—Christin Melton
The study was supported by Endo Pharmaceuticals Inc.
____________________________________________________________________________________________________________
Diclofenac Gel Relieves Pain and Stiffness in Osteoarthritis of the Hand
 In a randomized placebo-controlled trial, 6 weeks of treatment with diclofenac sodium 1% gel decreased joint pain and stiffness and improved function for more than 90% of adults with hand osteoarthritis (OA). Investigators, who presented the data during a poster session at the AAPM 28th Annual Meeting, said diclofenac was well tolerated by participants and caused no serious treatment-related events. Diclofenac is a topical nonsteroidal anti-inflammatory drug (NSAID). Like other NSAIDs, it does not modify the course of OA, but the authors said prior studies suggest relieving pain in patients with hand OA improves functional status.
In a randomized placebo-controlled trial, 6 weeks of treatment with diclofenac sodium 1% gel decreased joint pain and stiffness and improved function for more than 90% of adults with hand osteoarthritis (OA). Investigators, who presented the data during a poster session at the AAPM 28th Annual Meeting, said diclofenac was well tolerated by participants and caused no serious treatment-related events. Diclofenac is a topical nonsteroidal anti-inflammatory drug (NSAID). Like other NSAIDs, it does not modify the course of OA, but the authors said prior studies suggest relieving pain in patients with hand OA improves functional status.
The study enrolled 385 men and women 40 years or older who had mild to moderately painful OA affecting joints in their dominant hand for at least 1 year. During that time, participants were required to have treated OA pain with an NSAID at least once. Individuals with OA in joints outside the hand or with rheumatic diseases or fibromyalgia were excluded. Radiography revealed that 62.6% of eligible patients had OA affecting the first carpometacarpal (CMC-1) joints, which are located at the base of the thumb, as well as other joints in the hand. In 8.8% of patients, OA affected only the CMC-1 joints, whereas 28.6% of patients had OA confined to the intraphalangeal joints.
Following a 1-week washout period during which no analgesics were used, participants were randomized to diclofenac (n=198) or placebo (n=187). The assigned gel was applied in 2 g doses to each hand four times daily for 8 weeks. Neither participants nor investigators knew which patients received the active drug. Patients could supplement the gel with up to 4 g of oral acetaminophen daily for pain, provided they stopped using the pain reliever at least 36 hours prior to evaluation.
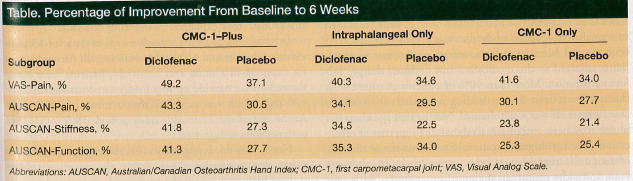
Pain intensity, stiffness, and function were measured at baseline and again at 6 weeks (Table) using the Visual Analog Scale (VAS) and the Australian/Canadian Osteoarthritis Hand Index (AUSCAN). VAS outcomes at 6 weeks showed significantly greater pain relief with diclofenac than with placebo, regardless of which joints were affected. AUSCAN, which assesses pain, stiffness, and function, found diclofenac superior to placebo across all three subscales in OA patients with CMC-1 plus additional joint involvement and in patients with intraphalangeal involvement. Diclofenac performed similarly to placebo on all measures in patients with OA confined to the CMC-1 joints. Because VAS and AUSCAN offered conflicting results regarding pain relief in the CMC-1–only group and AUSCAN failed to detect significant improvement for the CMC-1–only group alone, the authors concluded that AUSCAN “may not be a sensitive instrument for measuring pain in patients with OA limited to the thumb.”
The overall rate of adverse events was similar between the diclofenac arm and the placebo arm (52.0% vs 43.9%, respectively), with headache the most commonly reported adverse event in both arms (11.1% vs 10.2%, respectively). Gastrointestinal effects are a concern with oral NSAID use. Although the diclofenac group reported a higher rate of gastrointestinal events than the placebo arm (7.6% vs 3.7%, respectively), only two cases were thought possibly related to diclofenac use.—Christin Melton
This study was funded by Novartis Consumer Health.
______________________________________________________________________________________________________
Skeletal Muscle Mass Declines in Older Adults With Chronic Pain
Chronic nonmalignant pain in older adults correlates with reduced skeletal muscle mass (SMM) and impaired balance, according to a study presented during a poster session at the AAPM 28th Annual Meeting. Armando Miciano, MD, FAAPMR, Nevada Rehabilitation Institute, Las Vegas, who conducted the retrospective study, recommended geriatric patients who have chronic pain “focus on increasing strength and balance in order to decrease functional limitations.”
To compare SMM between community- dwelling older adults with chronic pain (n=26) and those without pain symptoms (n=23), Miciano relied on bioelectric impedance analysis (BIA). The noninvasive devices for BIA send low-level electric current through the body tissues (generally via electrodes placed at the wrist and ankle) and return an estimate of the individual’s total body water, which is then used to calculate muscle mass and adiposity. Chronic pain sufferers had a mean SMM of 20.94 kg (range, 11-40 kg) compared with 24.58 kg (range, 12-39 kg) for their pain-free counterparts.
Individuals in the chronic pain group completed the Pain Disability Questionnaire (PDQ) from the American Medical Association Guides to Evaluation of Permanent Impairment, 6th edition. This self-administered 15-question instrument asks patients to rate the effect of pain on their daily activities, income, medication use, and mood using a scale of 0 to 10, with 0 indicating no effect and 10 indicating maximum effect. Mean PDQ for participants in the chronic pain group was 59 points, which Miciano said reflects mild pain-related impairment. He found that a higher degree of pain-related impairment tended to correlate with lower SMM.
Function was evaluated for all participants using the 6-Minute Walk Test (6MWT), designed to measure exercise tolerance. Results from the 6MWT were used to produce a work performance percentage. The Berg Balance Scale (BBS), which is used to calculate fall risk based on the subject’s ability to perform a series of simple physical tasks, was also administered to everyone taking part in the study. BBS scores range from 0 to 56 points, with lower scores reflecting a greater risk of falling.
Although both cohorts’ mean BBS scores landed in the low fall risk range of 41 to 56 points, the chronic pain group had a significantly lower mean BBS score than the pain-free group (42 vs 49 points, respectively). The difference in mean 6MWT scores was not statistically significant between the chronic pain group and pain-free group (63.14% vs 64.42%, respectively), which suggests that chronic pain does not necessarily compromise one’s ability to exercise.
In addition to having lower SMM, individuals with chronic pain who experienced a greater degree of pain-related impairment demonstrated poorer physical function. Previous studies have demonstrated a link between low muscle mass and fall risk in older adults and support the idea that exercise—particularly strength-training—can increase SMM. Miciano therefore recommended patients with chronic pain exercise and work on increasing their strength to mitigate the limiting effects of chronic pain on function. He also called for additional study into the relationships between SMM, BBS, and work performance in older adults.
—Christin Melton


