Peer Reviewed
Acute Disseminated Encephalomyelitis
Authors:
Viola I. Winata, MD
Resident, Department of Pediatrics, Flushing Hospital Medical Center, Queens, New York
Firdous Rizvi, MD
Pediatric Neurologist, Jamaica Hospital Medical Center, Queens, New York
Dakshayani Guttal, MD
Director of Inpatients, Department of Pediatrics, Jamaica Hospital Medical Center, Queens, New York
Lily Q. Lew, MD
Vice Chair of Education and Research, Division of Pediatric Endocrinology, Flushing Hospital Medical Center, Queens, New York
Susana Rapaport, MD
Chairman, Department of Pediatrics, Flushing Hospital Medical Center, Queens, New York
Citation:
Winata VI, Rizvi F, Guttal D, Lew LQ, Rapaport S. Acute disseminated encephalomyelitis [published online December 11, 2017]. Consultant360.
A 4-year-old girl presented with vomiting for 2 days. She had had rhinorrhea, frontal headache, and watery eyes for 1 week prior to the vomiting, for which she had been treated with loratadine, 5 mg/d.
Presentation. She appeared tired and less-active than usual 1 day later. One day prior to the vomiting, she had received the measles, mumps, and rubella (MMR) and varicella vaccines. On the same day, she had a wobbly gait and was warm to the touch. Her temperature was not measured. There was no family history of a similar condition and no history of accidental drug or toxin ingestion. She had normal bladder and bowel control.
Physical examination. On physical examination, her weight was 19 kg (88th percentile), her height was 105 cm (79th percentile), her blood pressure was 96/59 mm Hg, her pulse rate was 70 beats/min, and she was afebrile. There were no signs of dehydration. Neurologic examination revealed a sleepy but arousable child with a Pediatric Glasgow Coma Score (PGCS) of 15 of 15. Cranial nerves II to XII were normal. Plantar reflex test results were positive for dorsiflexion of the big toes. Her gait was unstable. Finger-to-nose test results were normal, and no tremors were observed. Muscle strength and sensory function were normal. Abdominal examination revealed no hepatosplenomegaly. The rest of the physical examination findings were normal.
Laboratory tests. Laboratory test results included a normal complete blood cell count. Serum chemistry test results included the following values: sodium, 138 mEq/L; potassium, 4.3 mEq/L; chloride, 100 mEq/L; carbon dioxide, 23 mEq/L; glucose, 81 mg/dL; blood urea nitrogen, 10 mg/dL; creatinine, 0.4 mg/dL; alanine transaminase, 21 U/L; calcium, 10 mg/dL; alkaline phosphatase, 189 U/L; total protein, 6.9 g/dL; and albumin, 3.7 g/dL. The C-reactive protein level was 0.6 mg/dL. QuantiFERON test results were negative for Mycobacterium tuberculosis infection, and results of urine toxicology tests were negative for drugs or other toxins.
Computed tomography scans of the brain revealed patchy hypodensity in the periventricular and subcortical white matter. Magnetic resonance imaging (MRI) showed extensive periventricular and subcortical white matter T2 hyperintensities with multiple foci of abnormal enhancement in the corpus callosum, optic nerves, optic chiasm, optic tracts, basal ganglia, and internal capsule, suggestive of a demyelinating process or vasculitis (Figure). Cerebrospinal fluid (CSF) analysis showed no cells. CSF was sent for culture, chemistries, bacterial antigen testing, herpes simplex virus 1 and 2 polymerase chain reaction assay, varicella-zoster virus IgM antibody testing, myelin basic protein testing, protein electrophoresis, and oligoclonal band screening.
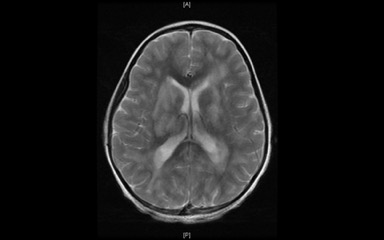
Figure. Axial T2 MRI image of the brain demonstrating extensive periventricular hyperintensities.
She was admitted with a presumptive diagnosis of acute disseminated encephalomyelitis (ADEM). Methylprednisolone, 20 mg/kg/d, and ceftriaxone, 100 mg/kg/d, were started. Following the first dose of corticosteroid, she developed slurred speech and difficulty pronouncing words and was somnolent but still arousable. Her PGCS score was 12 of 15. Her heart rate was irregular, ranging from 55 to 130 beats/min, and her blood pressure fluctuated (systolic, 80-106 mm Hg; diastolic, 40-69 mm Hg). An electrocardiogram revealed sinus arrhythmia.
The dose of methylprednisolone was increased to 30 mg/kg/d, and intravenous vancomycin and intravenous acyclovir were started. On day 2 of the high-dose corticosteroid, she became fully alert, with a PGCS score of 15 of 15. Her gait remained unstable, and physical therapy was started. After 5 days of intravenous methylprednisolone, all of her neurologic symptoms and signs resolved.
She was discharged on hospital day 8 with no neurologic deficits. Results of all blood and CSF tests were subsequently reported as normal.
Differential diagnosis. The differential diagnosis at presentation and prior to neurologic imaging included central nervous system (CNS) infection, intracranial mass, drug or toxin ingestion, and a demyelinating disorder. There was no history of drug or toxin ingestion, which was confirmed by negative urine drug toxicology results. No specific infectious agent was identified in the CSF, blood, or urine. The brain imaging studies did not show an intracranial mass lesion. The MRI of the brain showed typical findings of ADEM.
Discussion. ADEM is an inflammatory demyelinating disease marked by multifocal neurologic deficits. It can occur at any age, predominantly between 5 and 8 years. The current theory is that the neurologic insult is due to an autoimmune process triggered by a preceding infection or vaccination.1-4
This rare condition has an incidence ranging from 0.07 to 0.4 per 100,000 pediatric population per year and occurs mostly in winter and spring months with no specific ethnic distribution.2,5 Antecedent infection could be identified in 70% of cases, and 5% to 10% of cases occurred after vaccination.6-8
The clinical outcome is difficult to predict. It can range from full recovery to death. The reported rate of full recovery is from 57% to 89%, and some are left with residual motor and/or cognitive deficits.2,3
Our patient met the ADEM diagnostic criteria based on the revised guidelines of the International Pediatric Multiple Sclerosis Study Group (IPMSSG).2,9 This was our patient’s first polyfocal clinical neurologic event with presumed inflammatory cause. She had encephalopathy that cannot be explained by fever. She had typical brain MRI abnormalities showing diffuse, poorly demarcated, large (>1-2 cm) lesions predominantly in the cerebral white matter, deep gray-matter lesions (in the thalamus or basal ganglia), and no T1 hypointense white matter lesions. There was no new clinical or MRI finding 3 months or more after the onset.
ADEM can initially present with lethargy, fever, headache, vomiting, and meningeal signs. As the encephalopathy evolves, symptoms can range from ongoing confusion or irritability to coma.2 Many patients with encephalopathy present with a combination of acute symptoms such as seizure, autonomic instability, and respiratory irregularity that require priority in management. Evaluation and treatment for infectious or parainfectious conditions are necessary until the specific diagnosis is confirmed.3
Infections reported in association with ADEM include influenza, Epstein-Barr virus, cytomegalovirus, varicella virus, enterovirus, measles, mumps, rubella, herpes simplex virus, and Mycoplasma pneumoniae. ADEM can occur after immunization for rabies, MMR, Japanese encephalitis virus, pertussis, diphtheria-polio-tetanus, and influenza.2 The incubation period between the preceding infectious illness or vaccination and the neurologic symptoms can range from 2 to 30 days.3 In our patient, ADEM might have been triggered by the combination of preceding viral illness and vaccinations. However, it usually takes a few days for the symptoms of a demyelinating disorder to appear after immunization (with a mean of 14 days).8
Our patient’s brain MRI showed lesions typical for ADEM—specifically, extensive periventricular and subcortical T2 hyperintensities with multiple foci of abnormal enhancement located at corpus callosum, optic nerves, optic chiasm, optic tracts, basal ganglia, and internal capsule (Figure).2,3 Brain MRI is the diagnostic imaging study of choice for ADEM.2,3
Lumbar puncture test results were diagnostic for encephalitis. In ADEM, the CSF findings may be similar to those found in infectious encephalitis or meningitis.3 Consequently, treatment with antibiotics and antiviral medications is necessary until the results of the CSF studies become available.
High-dose methylprednisolone (20-30 mg/kg/d for 5 days) is the recommended treatment. Accordingly, our patient’s methylprednisolone dose was increased to 30 mg/kg/d for a total of 5 days. She improved dramatically, recovered within 6 days, and was discharged with a tapering corticosteroid dose. In cases of poor response to treatment with corticosteroids, alternative therapies include intravenous immunoglobulin, 2 g/kg for 2 to 5 days, and plasmapheresis.1-4
In most patients, the course of ADEM usually is monophasic with fluctuating symptoms and MRI findings up to 3 months.9 Relapses can occur in 5% to 25% of cases 3 months or more after the first onset of ADEM.10 The relapsing forms can be divided into recurrent (80%) if there is a new ADEM event with a recurrence of the initial ADEM signs and symptoms, and multiphasic (20%) if there is a new ADEM event with different signs and symptoms from the first one and involving a new anatomic area confirmed by history, neurologic examination, and neuroimaging.9,10 In the revised IPMSSG guidelines, the timing from completing the coricosteroid treatment to the relapse course is no longer pertinent as a diagnostic criterion.2,9 However, we need to acknowledge that ADEM symptoms can recur during the oral corticosteroid taper or after completion of the taper.
In a prospective study of a 9-year follow-up of children with ADEM, the risk of developing multiple sclerosis (MS) from ADEM was found to be 6%.11 Differentiation between ADEM and MS continues to be challenging, especially with multiphasic ADEM when demarcation between ADEM relapse and MS becomes unclear. Relapse after a second encephalopathic episode suggests a chronic disorder when MS needs to be considered. In this scenario, ADEM becomes the first symptom of MS.
First-onset ADEM differs from MS in that ADEM features impairment in mental status, CSF pleocytosis, absence of oligoclonal bands, involvement of CNS gray matter, and the presence of fever.8
Conclusion. ADEM is an uncommon acute neurologic condition that can be diagnosed with a careful history, appropriate laboratory evaluations, and MRI of the brain. Patients with ADEM need to have continued clinical and MRI follow-up to screen for early MS.
References:
- Khurana DS, Melvin JJ, Kothare SV, et al. Acute disseminated encephalomyelitis in children: discordant neurologic and neuroimaging abnormalities and response to plasmapheresis. Pediatrics. 2005;116(2):431-436. doi:10.1542/peds.2004-2038
- Ness JM. Demyelinating disorders of the central nervous system. In: Kliegman RM, Stanton BF, St. Geme J III, Schor NF, eds. Nelson Textbook of Pediatrics. Vol 2. 20th ed. Philadelphia, PA: Elsevier; 2015:2919-2925.
- Falchek SJ. Encephalitis in the pediatric population. Pediatr Rev. 2012;33(3):122-133.
- Murthy SNK, Faden HS, Cohen ME, Bakshi R. Acute disseminated encephalomyelitis in children. Pediatrics. 2002;110(2 pt 1):e21. doi:10.1542/peds.110.2.e21
- Lotze TE, Chadwick DJ. Acute disseminated encephalomyelitis in children: pathogenesis, clinical features, and diagnosis. UpToDate. https://www.uptodate.com/contents/acute-disseminated-encephalomyelitis-in-children-pathogenesis-clinical-features-and-diagnosis. Updated March 16, 2017. Accessed December 6, 2017.
- Hynson JL, Kornberg AJ, Coleman LT, Shield L, Harvey AS, Kean MJ. Clinical and neuroradiologic features of acute disseminated encephalomyelitis in children. Neurology. 2001;56(10):1308-1312.
- Tenembaum S, Chamoles N, Fejerman N. Acute disseminated encephalomyelitis: a long-term follow-up study of 84 pediatric patients. Neurology. 2002;59(8):1224-1231.
- Karussis D, Petrou P. The spectrum of post-vaccination inflammatory CNS demyelinating syndromes. Autoimmun Rev. 2014;13(3):215-224. doi:10.1016/j.autrev.2013.10.003
- Pena JA, Lotze TE. Pediatric multiple sclerosis: current concepts and consensus definitions. Autoimmune Dis. 2013;2013. doi:10.1155/2013/673947
- Marchioni E, Tavazzi E, Minoli L, et al. Acute disseminated encephalomyelitis. Neurol Sci. 2008;29(suppl 2):286-288. doi:10.1007/s10072-008-0966-6
- Mar S, Lenox J, Benzinger T, Brown S, Noetzel M. Long-term prognosis of pediatric patients with relapsing acute disseminated encephalomyelitis. J Child Neurol. 2010;25(6):681-688. doi:10.1177/0883073809343320


