Lies, Damned Lies, and COPD
Author:
Samuel Louie, MD
Citation:
Louie S. Lies, damned lies, and COPD. Consultant. 2019;59(10):300-307, 310.
Vladimir Lenin, during his reign over the Soviet Union, asserted (perhaps apocryphally), “A lie told often enough becomes the truth.” For decades, clinicians and professors in medical schools declared that chronic obstructive pulmonary disease (COPD) is “irreversible,” but this lie and stereotype is disproved by evidence of bronchodilator reversibility (forced expiratory volume in the first second of expiration, or FEV1) in nearly 66% of patients with moderate to very severe COPD.1
Are clinicians still relying on bronchodilator reversibility to diagnose asthma and exclude COPD? Why do we wait to recognize and treat COPD until after patients have lost significant lung function? Why do we treat asthma in patients with normal lung function despite that no treatment, including inhaled corticosteroids (ICSs), alters the natural course of the disease?
Here I offer a critical perspective from one clinician’s humbling experience learning from patients with COPD since 1993.
COPD TODAY AND YESTERDAY
November is National COPD Awareness Month; the Global Initiative for Chronic Obstructive Lung Disease (GOLD) has issued reports since 2001; the COPD Foundation was established in 2004; and the COPD National Action Plan was released in 2017. Yet COPD remains the fourth leading cause of death in the United States, after only heart disease, cancer, and unintentional injuries.2 COPD is the fourth leading cause of disability3 and the second leading cause of reduced disability-adjusted life years, trailing only ischemic heart disease.4
The term chronic obstructive pulmonary disease (COPD) was first used nearly 55 years ago to include asthma, chronic bronchitis, and bronchiectasis.5,6 Not everyone was enamored with the term, which groups older men into the “pink puffers” (emphysema) together with the “blue bloaters” (chronic bronchitis). These two COPD phenotypes represent different, final stages of obstructive lung diseases. COPD phenotypes today include nonexacerbator, exacerbator with emphysema, exacerbator with chronic bronchitis, and asthma-COPD overlap.7
My clinical experience argues that COPD is a childhood disorder caused by cigarette smoking with old-age consequences. COPD manifests in younger adults, with more than 50% of new COPD diagnoses being made in persons younger than 65.8
The emerging truth is that COPD is not a single disease but is a collection of progressive, debilitating lung disease phenotypes distinguished by difficulty breathing, cough, and phlegm production with or without acute exacerbations that is often but not necessarily associated with cigarette smoking. A psychological pitfall accompanies a newly diagnosed patient with COPD compared with asthma. Asthma tends to elicit empathy, because many believe it is an inherited and reversible disease exacerbated by environmental triggers. In my experience, COPD is met instead with indifference and even disdain, because it is assumed to be caused by nicotine addiction, and canon says that COPD is an irreversible obstructive airway disease. The irony here is that both asthma and COPD are chronic diseases without a cure, but having asthma is far more acceptable in society.
The diagnosis of COPD today is more difficult, driven in part by the recognition of asthma-COPD overlap syndrome (ACOS),9 the European Respiratory Society/American Thoracic Society definition of severe asthma,10 and non–cystic fibrosis-related bronchiectasis overlapping with COPD.
There is general acceptance of the term COPD by the World Health Organization (WHO) and the GOLD report to describe a preventable and treatable chronic lung disease in adults, primarily occurring in tobacco cigarette smokers.4 Several excellent reviews of COPD care have been published,9 but I am generally disappointed by their accord with their view of COPD and published guidelines’ recommendations.
Only 38% of the nearly 16 million US adults with COPD are current smokers.11 Up to 25% of adults can develop COPD without ever having smoked a cigarette. Between 3% and 5% of those with COPD are adults between 20 and 50 years of age who have genetic panlobular basal emphysema from α1-antitrypsin deficiency (ATTD). GOLD recommends that all patients with a diagnosis of COPD be screened for ATTD. Patients with ATTD are typically younger than 45 years. A low concentration of α1-antitrypsin (<20% of normal) is highly suspicious for homozygous deficiency, and emphysema can progress.
The immunology of COPD is an area of controversy, but tobacco smoke compromises innate and adaptive immune responses that are important in preventing respiratory infections. Immune dysfunction in COPD has been blamed for disease severity and progression despite smoking cessation, as well as for acute exacerbations.12,13 Other important risk factors include older age, comorbidities such as gastroesophageal reflux disease (GERD), and a previous history of acute COPD exacerbations.14
“DO YOU WANT TO QUIT SMOKING?”
Mark Twain declared, “Giving up smoking is the easiest thing in the world. I know because I’ve done it thousands of times.” Smoking tobacco is the leading cause of preventable death, disease, and disability in the United States.15 What is a smoker? The Centers for Disease Control and Prevention (CDC) and the National Health Interview Survey define 3 important types16:
- Current smoker: An adult who has smoked 100 cigarettes in his or her lifetime and who currently smokes cigarettes. Beginning in 1991, this group was divided into “every-day” smokers or “some-days” smokers.
- Former smoker: An adult who has smoked at least 100 cigarettes in his or her lifetime but who has quit smoking.
- Never smoker: An adult who has never smoked, or who has smoked fewer than 100 cigarettes in his or her lifetime.
Smoking cessation is the only disease-modifying intervention that can prevent COPD, improve lung function, stop the long-term decline in FEV1, improve symptoms of COPD, and reduce the risk of acute exacerbations of COPD (AECOPD). Smoking cessation must be regarded as the most essential primary—if not lifesaving—therapeutic intervention in COPD, with other treatments such as bronchodilators important but ancillary to improving patient safety and health-related quality of life (HRQoL). To date, no conclusive clinical trial evidence exists demonstrating that ancillary treatments (including long-acting bronchodilators, ICSs, roflumilast, exercise, and oxygen therapy) even in combination modify the long-term decline in FEV1.
Other contributing risk factors such as air pollution, recurring lung infections from viruses and bacteria, asthma, poor socioeconomic conditions, and genetics pale in comparison to the dangers of smoking tobacco.
Cigarette smoking is responsible for more than 480,000 US deaths annually, including more than 41,000 deaths from secondhand smoke.15 An estimated 21% of adults and 24% of adolescents are current smokers.17,18 On average, smokers die 10 years earlier than never smokers.18
Results from the 2015 National Health Interview Survey of 33,672 adults found that 68% of current smokers want to quit and that 55% of current and former smokers had attempted to quit within the past year.19 The combination of counseling plus a smoking cessation pharmacological aid (nicotine replacement, varenicline, or bupropion) is more effective than either measure alone when making a quit attempt successful.20 Nevertheless, it is estimated that only 5% of smokers received both counseling and a pharmacological aid for smoking cessation.19
The Food and Drug Administration (FDA) removed the boxed warning about possible adverse neuropsychiatric effects of varenicline and bupropion in 2016 after the EAGLES study found varenicline to be more effective than placebo, the nicotine patch, and bupropion in helping smokers achieve abstinence, whereas bupropion and the nicotine patch were more effective than placebo.21 The benefits of smoking cessation generally outweigh the risk associated with these drugs. Clinicians should monitor patients for adverse neuropsychiatric effects, especially patients with a history of depression, anxiety, or schizophrenia.
Quitting smoking is beneficial to health at any age and to all persons with COPD, and cigarette smokers who quit before age 35 years have mortality rates similar to those who never smoked.19
The worst pitfall is to forego helping smokers stop smoking because they are neither ready nor want to. It is imperative for clinicians to avoid judging smokers. Approach smokers as an educator and advocate. I share with my patients the potential lifetime benefits of stopping and provide a timeline (Table 1).22

In a prospective study of 34,439 male British doctors from 1951 to 2001, smoking cessation at age 60, 50, 40, or 30 years gained, respectively, approximately 3, 6, 9, or 10 years of life expectancy compared with men who continued to smoke.23 Expect many smokers to try and fail to achieve abstinence, but also expect just as many smokers to succeed in stopping. The benefits are lifesaving.
A CLEAR AND PRESENT DANGER
Every 4 minutes, a patient dies from COPD.24 Every 45 seconds, a COPD patient will be admitted to hospital for an acute exacerbation.25 More than 160,000 people each year die from COPD (including asthma) in the United States.26 In 2016, COPD was the third leading cause of years of life lost and disability-adjusted life years in the United States.26
Warnings and public access to information from the WHO, the CDC, the National Institutes of Health, and health care experts have not improved the diagnosis of COPD or care of patients with COPD. The explanation may reside in the fact that both patients and clinicians unintentionally neglect COPD. Patients and clinicians may both misunderstand or minimize symptoms of COPD. GOLD has cited misperception and denial by patients as significant barriers to diagnosis, treatment and management.4
Smokers and nonsmokers may not complain to a clinician because they have become accustomed to or adapted to their symptoms, feeling “fine” when they are symptomatic when challenged, for example, by walking. Patients with COPD have taught me that the loss of lung function (FEV1) is “painless”—that is, they do not “feel” the loss. Patients with COPD often limit physical activities such as walking or climbing to avoid feeling distressed from dyspnea on exertion. It is important to ascertain whether patients engage in physical and social activities such as walking. Many patients with COPD attribute their symptoms of shortness of breath to aging or to lack of exercise, or they fear lung cancer as the cause of their chronic cough.27,28 “It is a smoker’s cough—it’s normal.” “It is harder to get around and to climb up the steps—I must be getting old and out of shape.” “No need for medicines. I just need to rest more.” If only the loss of lung function caused genuine pain!
Approximately 16 million Americans have received a diagnosis of COPD, but at least another 12 million remain undiagnosed and untreated.29-31 More than 50% of adults with abnormal pulmonary function test results are unaware that they meet the criteria for the diagnosis of COPD.32 Lung function can be reduced by more than 60% by the time COPD is diagnosed.33
Two nationally representative US National Health and Nutritional Examination Surveys demonstrate that despite apparent spirometry-confirmed obstruction, more than 70% of Americans aged 20 to 79 years did not receive a diagnosis of asthma or COPD, and that this high rate of underdiagnosis has not changed over 2 decades despite the GOLD report.34
Patients with COPD have become a daily test of my character and empathy. Every clinician can take this test: Do you confirm and recognize the diagnosis of COPD with spirometry? Then do you attempt to phenotype and endotype but avoid stereotyping the patient? And finally, do you conduct a “clinical trial of one” with an action plan to help patients begin their journey to reduce symptoms and reduce acute exacerbations living with COPD?4
Office spirometry remains severely underutilized in clinical practice.35,36 Using a retrospective cohort design and data from the Veterans Health Administration, investigators found that in some regions of the country, the rates of patients who had received a new diagnosis of COPD between 2003 and 2004 undergoing spirometry were surprisingly low, ranging from a high of 47.8% in Southern California to a low of 23% in Texas.37 Spirometry may be difficult to interpret for primary care providers.38,39 Asthma and COPD may be confused, since they can both exhibit fixed airflow obstruction.40
FEV1 from spirometry correlates with HRQoL, health care resource utilization, frequency of AECOPD, small-airway wall thickness, and mortality. FEV1 does not correlate with dyspnea, exercise tolerance, emphysema, static lung hyperinflation, body mass index (BMI), or skeletal muscle wasting.
To make matters worse, COPD is often misdiagnosed as asthma. A primary care provider may first incorrectly diagnose more than 50% of those affected with COPD with asthma instead.40 COPD symptoms in women are most commonly misdiagnosed as asthma.41
Despite nearly 20 new drug therapies for COPD having been FDA-approved between 2000 and 2018, the 30-day hospital readmission rate for COPD in the United States is unchanged, with 1 in 5 patients with COPD who are hospitalized for acute exacerbations readmitted within 30 days.42,43 Most patients with COPD do not receive maintenance drug therapies: 66.3% on commercial insurance and 70.9% on Medicare are undertreated despite carrying a diagnosis of COPD.44
It is a pitfall to think that COPD is a disease of men only, and that they are more prone than women to morbidity and mortality from COPD. More than half of US patients with COPD are women, and approximately half of COPD cases are in patients younger than 65 years of age,45 and between the ages of 45 and 64.46
COPD has killed more US women than men since 2000.47 In 2017, chronic lower respiratory diseases, primarily COPD, were the third leading cause of death among US women.47 Death rates from COPD have fallen among US men but remain unchanged for women. More US women than men are also living with COPD.48 Explanations for this gender disparity include delayed or later diagnosis of COPD in women, when COPD is more advanced and treatment less effective.49 Women who smoke tend to develop COPD at a younger age, with fewer pack-years.49
COPD IS A SYNDROME
COPD is a syndrome and not a single disease (Figure). It can be recognized by exercise intolerance and exacerbations in a smoker, whether current, former, or never.

Figure. COPD is a syndrome. Reproduced with permission from: Louie S, Zeki AA, Schivo M, et al. The asthma–chronic obstructive pulmonary disease overlap syndrome: pharmacotherapeutic considerations. Expert Rev Clin Pharmacol. 2013;6(2):197-219.9
COPD is a childhood disorder with old-age consequences. The average age at which a person begins smoking tobacco is 15.3 years.15 Tobacco dependence very often continues into adulthood. Nearly 90% of adult smokers began smoking before the age of 18.50
Not 15%, not 20%, but 50% of smokers develop COPD!51,52 One of every 2 smokers can develop COPD if they continue smoking!
Like asthma, COPD is complicated by misclassification and the absence of consensus about the definition, and the fact that asthma and COPD can overlap. Instead of chronic bronchitis and emphysema, the GOLD definition of COPD is descriptive of a clinical state.4
Of all the different versions of the GOLD definition of COPD, I prefer the original one from 2001, which states that “COPD is a disease state characterized by airflow limitations that is not fully reversible. The airflow limitation is usually progressive and associated with an inflammatory response of the lung to noxious particles or gases.”53
GOLD later added systemic consequences to this definition. The definition of COPD from GOLD 2019 is “a common, preventable and treatable disease that is characterized by persistent respiratory symptoms and airflow limitation that is due to airway and/or alveolar abnormalities usually caused by significant exposure to noxious particles or gases.”4
Neither definition recognizes the significant latent period between the early stages of “pre-COPD” or chronic bronchitis (postbronchodilator FEV1/FVC ³0.70) and clinical recognition of COPD with spirometry (postbronchodilator FEV1/FVC <0.70) when patients will have lost 60% of their lung function.33
The reasons for the latent period and the delay in COPD diagnosis is not entirely clear, but the relief of symptoms by resting or curtailing exercise and other physical activities that cause symptoms must be a major reason for why patients wait and wait for help. Loss of lung function (FEV1) may be painless.
Table 2 outlines some of the lies and the truths about COPD.

GUIDELINES DO NOT CARE FOR PATIENTS
Guidelines do not care for patients; clinicians with empathy and professional experience do. Guidelines such as the GOLD 2019 strategy can be valuable, since they summarize key information from clinical trials and population-based studies concerning a disease. However, all semblances of patients’ individuality are removed statistically, and confidence intervals are crunched out. Expert opinion is rendered on statistical analyses, and recommendations for treatment of a disease are produced. But guidelines do not concern themselves with or take care of individual patients.
It is a fallacy to think that strict adherence to evidence-based COPD treatment algorithms derived from clinical trials is necessary to treat properly a patient with a disease. Clinicians must become more flexible, avoid a “prescribe and forget” attitude, and embrace the opportunity to conduct a “clinical trial of one” in every patient.
Since 2001, the GOLD guidelines have promoted COPD as a single disease, enough to make recommendations for treatment and management despite the presence of clinical heterogeneity. The appearance of the 2019 GOLD guidelines is a dramatic departure from the past, where treatment and management are divided into an initial encounter and follow-up visits to review, assess, and adjust treatment based on the burden of symptoms and/or frequency of AECOPD. The resemblance to the asthma GINA 2019 guidelines for asthma is unmistakable.54 Clinicians can no longer prescribe and forget to reassess in COPD patients.
TREATABLE TRAITS IN COPD
In the GOLD revised assessment scheme,4 patients should undergo spirometry to confirm the diagnosis of COPD and determine the severity of airflow limitation and, therefore, their GOLD grade or stage (1, 2, 3, or 4). They should also undergo assessment of treatable traits: symptoms and exacerbations. Assess dyspnea using the modified Medical Research Council (mMRC) dyspnea scale (available at https://www.mdcalc.com/mmrc-modified-medical-research-council-dyspnea-scale) or symptoms using the COPD Assessment Test (CAT) (available at https://www.catestonline.org/patient-site-test-page-english.html). Finally, their history of exacerbations (including prior hospitalizations) should be recorded.
At UC Davis, we find the CAT score (0-40) very helpful in the hospital and clinics, as it is available in 57 other languages in addition to English and immediately brings the patient with COPD into the clinical setting. CAT scores of less than 10 have a low impact on health status, 10 to 20 a medium impact, 21 to 30 a high impact, and greater than 30 a very high impact, requiring gradually more intervention. A CAT score greater than 20 indicates that severe symptoms of breathlessness are present.
The GOLD grade (1, 2, 3, or 4) provides information regarding severity of airflow limitation from spirometry, while the letter (A, B, C, or D) provides information regarding symptom burden and risk of exacerbation, which can be used to guide initial pharmacotherapy.4
Classification of airflow limitation severity in COPD is based on postbronchodilator FEV1. Instead of the GOLD grade 1 (mild), 2 (moderate), 3 (severe), and 4 (very severe), I propose the following revision of GOLD to reflect my clinical experience:
- GOLD 1—early: FEV1 ³ 80% predicted
- GOLD 2—late: 50% £ FEV1 < 80% predicted
- GOLD 3—too late: 30% £ FEV1 < 50% predicted
- GOLD 4—way too late: FEV1 < 30% predicted
This change raises the call to earlier action on both the clinician and the patient with COPD and their family. The adjective moderate may delay interventions such as smoking cessation, bronchodilators, and pulmonary rehabilitation. Any patient with symptoms of dyspnea, cough, and sputum production should benefit from smoking cessation, exercise, and bronchodilators for chronic bronchitis before the FEV1/FVC postbronchodilator becomes less than 0.70.
The results of the 3-year observational study ECLIPSE (Evaluation of COPD Longitudinally to Identify Predictive Surrogate Endpoints)55 suggested an early association between declining lung function (GOLD stage by FEV1 % predicted) and the frequency of acute exacerbations and hospitalizations based on GOLD grades 2 to 4 that may not be detected with the ABCD scheme without spirometry. The frequent-exacerbation phenotype is best discovered by recall of previous AECOPD in the past 12 months.55 As COPD progresses and FEV1 declines (£50% predicted), the frequency and severity of exacerbations can be expected to increase.
The ABCD scheme is used by GOLD to recommend initial pharmacological treatment.4 An effort to understand the relationship of COPD groups A, B, C, and D to symptoms and acute exacerbations, including hospitalization, remains controversial with each new version of the GOLD grading system. The grading system is linked—for better or for worse—to evidence-based pharmacological treatment recommendations by the GOLD report.
A common pitfall is to believe that bronchodilator reversibility is a prerequisite for bronchodilators to be prescribed and for patients to respond. Prescription of bronchodilators is based on the presence of treatable traits, not reversibility. The treatable traits are symptoms assessed by CAT or mMRC and exacerbations assessed by the frequency of them. The two goals of COPD management are to reduce symptoms, including improvement in exercise tolerance and health status, and to reduce risks, including preventing progression, exacerbations, and mortality.
Frequent follow-up visits within 2 to 6 weeks are needed for review, assessment, and adjustment to implement a personalized management strategy predominantly based on the evaluation of symptoms burden (as assessed with CAT) and future risk of exacerbations. Ascertain by history and review of the electronic medical record that there were no urgent care visits, emergency department visits, or hospitalizations.
The first follow-up visit is very important to discover further aspects of the patient’s COPD phenotype—for example, the presence or absence of peripheral blood eosinophilia (eosinophil count ³300 cells/µL) and any difficulties or barriers to treatment that have arisen since the initial visit, such as poor inhaler technique, suboptimal peak inspiratory flow rates, medication nonadherence, and the cost of drugs. Important comorbidities that must be addressed include gastroesophageal reflux disease, obstructive sleep apnea, heart disease, and asthma.
Every patient with COPD must be able to demonstrate proper technique with their inhaler device, including metered-dose inhaler (MDI), dry-powder inhaler (DPI), soft-mist inhaler, or nebulizer system. All inhalers and nebulizer systems are FDA-approved and effective, but their ability to deliver drugs into the lungs requires education and repeated demonstration. Switching inhaler devices or the drug molecule(s) should be considered if the goals of management are not being met.
Pharmacological treatments aimed to reduce breathlessness and/or acute exacerbations are escalated or deescalated during follow-up visits depending on the cycle of review, assessment, and adjustment.4
THE CLINICAL TRIAL OF ONE
The patient-provider partnership is vital in a comprehensive, patient-centric COPD care program. The clinician must engage the COPD patient and remember empathy is not an occupational hazard. Experience taught me virtually every patient needs more COPD disease-state education, inhaler device training, safety awareness, and a personalized written COPD action plan to fully participate in the quest to control symptoms and reduce the risk of AECOPD.
Each patient is told that their response to maintenance or controller drug therapy can direct changes in treatment and management to better individualize their care. This is the “clinical trial of one.”
I tell patients with COPD during their first visit or during a hospitalization for AECOPD that they have a preventable, treatable, and partially reversible disease called COPD. Then I tell them that they have “chronic air constipation.” This phrase is easier to understand and translate than static lung hyperinflation. Dynamic lung hyperinflation is “acute air constipation.”
As COPD progresses, patients may develop lung hyperinflation. Several changes occur, including loss of lung elastic recoil, alveolar and parenchymal tissue destruction, airway narrowing, and small airway inflammation. These changes will affect a patient’s ability to exhale, and progressively more air will be trapped in the lung, resulting in pulmonary hyperinflation or what we have called “chronic air constipation.”
As COPD progresses, hyperinflation of the lungs becomes more pronounced.56 Hyperinflation leads to impairment of respiratory muscle function and gas exchange and increases work of breathing.57 During exercise, hyperinflation worsens; this further reduces exercise tolerance in patients with COPD. Treatment with bronchodilators improves hyperinflation, reduces the work of breathing, and improves exercise tolerance.58
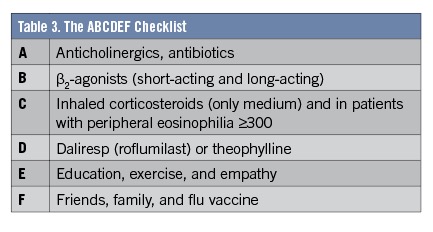
The ABCDEF checklist developed at UC Davis can help clinicians and patients recall the order of COPD drug treatments targeting the treatable traits symptoms and exacerbation risk, as well as nonpharmacological interventions, including exercise, vaccinations, and friends (Table 3). Smoking cessation, pulmonary rehabilitation, vaccinations, and palliative care all contribute to improving the health and safety of patients with COPD.
CONSULTATION
COPD is underdiagnosed and consequently undertreated, but even when the clinical diagnosis has been confirmed by spirometry, patients with COPD are not prescribed or use evidence-based maintenance therapy to reduce symptoms and exacerbations. It is wrong to wait to diagnose COPD until people have lost 60% of their lung function. We must fight indifference!
Clinicians can diagnose chronic bronchitis or “pre-COPD” if the FEV1/FVC is 0.70 or greater in a smoker with chronic cough, sputum production, and dyspnea. Clinicians can engage and educate patients to stop smoking tobacco earlier at a younger age when it can make the greatest impact on mortality, and we can promote exercise.
The terms asthma and COPD color our clinical judgment with stereotypes. These diagnostic labels are even less helpful with the recognition of ACOS. Treat each patient individually based on their phenotype and endotype, avoid stereotyping, and improve patient safety and health with lifestyle changes and selected drug therapy.
Key aspects of consultation at UC Davis for COPD include first confirming the diagnosis, then engaging the patient to assess their understanding of COPD (ie, chronic air constipation), including what has worked and what has not worked to help them breathe better. The patient takes the CAT, and the results are explained with the patient without casting social judgment on their debility, both physical and psychological. We avoid stereotyping and attempt to phenotype and endotype the patient’s COPD as eosinophilic or noneosinophilic, and order tests in search for a component of asthma.
Next, we boost their education quickly on the goals of smoking cessation and COPD management to reduce symptoms and to reduce risks from exacerbations. An individualized and daily COPD action plan is designed and written to summarize the consultation. No intervention is more vital than smoking cessation, which can stop progression of COPD. Vaccinations are encouraged for both pneumococcal pneumonia and influenza.
Spirometry, flow-volume loops, diffusing capacity for carbon monoxide (if available), and chest radiographs or chest computed tomography scans are reviewed with an eye to lung nodule(s) and bronchiectasis in addition to pulmonary emphysema.
We ascertain the patient’s capability or competence using his or her prescribed albuterol rescue inhaler and controller medications contained in different inhaler device designs (eg, MDI, DPI, soft mist) or resort to a traditional jet nebulizer or closed nebulizer system. Atrocious inhaler technique is to be expected in the great majority of patients with COPD.59
The ABCDEF tool is employed to explain the rationale for a clinical trial of one. This tool draws the patient with COPD into decision-making and allows him or her to anticipate the next treatment(s). Only if the presentation is atypical or if the patient reports excessive mucus production do we consider ordering chest CT scan to look for bronchiectasis, emphysema, lung nodule(s), or interstitial lung disease, which can accompany COPD concurrently.
Finally, the benefits of pulmonary rehabilitation are introduced as the centerpiece of our chronic disease management program for all patients with COPD.60,61
I cannot do better than to conclude with adaptation of the 1963 remarks of Claude A. Frazier, MD, about asthma that I quoted previously in Consultant62: “COPD is a problem disease; its capricious nature defies control and keeps the health of the COPD patient always in a precarious state. And, improper treatment can make the COPD worse. Each person is an individual and rules change with individuals.”63
Samuel Louie, MD, is a professor of medicine in the Department of Internal Medicine, Division of Pulmonary, Critical Care, and Sleep Medicine and is the director of the UC Davis Asthma Network (UCAN) and Reversible Obstructive Airways Diseases (ROAD) Center at UC Davis Health in Sacramento, California.

REFERENCES:
- Tashkin DP, Celli B, Decramer M, et al. Bronchodilator responsiveness in patients with COPD. Eur Respir J. 2008;31(4):742-750.
- Kochanek KD, Murphy S, Xu J, Arias E. Mortality in the United States, 2016. NCHS Data Brief. 2017;(293):1-8.
- Centers for Disease Control and Prevention (CDC). Prevalence and most common causes of disability among adults—United States, 2005. MMWR Morb Mortal Wkly Rep. 2009;58(16):421-426.
- Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease 2019 Report. Global Initiative for Chronic Obstructive Lung Disease. https://goldcopd.org/wp-content/uploads/2018/11/GOLD-2019-v1.7-FINAL-14Nov2018-WMS.pdf. Accessed September 26, 2019.
- Petty TL. The history of COPD. Int J Chron Obstruct Pulmon Dis. 2006;1(1):3-14.
- Briscoe WA, Nash ES. The slow space in chronic obstructive pulmonary disease. Ann N Y Acad Sci. 1965;121(3):706-722.
- Sidhaye VK, Nishida K, Martinez FJ. Precision medicine in COPD: where are we and where do we need to go? Eur Respir Rev. 2018;27(149):180022.
- Centers for Disease Control and Prevention. Chronic obstructive pulmonary disease among adults—United States, 2011. MMWR Morb Mortal Wkly Rep. 2012;61(46):938-943.
- Louie S, Zeki AA, Schivo M, et al. The asthma–chronic obstructive pulmonary disease overlap syndrome: pharmacotherapeutic considerations. Expert Rev Clin Pharmacol. 2013;6(2):197-21
- Chung KF, Wenzel SE, Brozek JL, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343-373.
- Wheaton AG, Cunningham TJ, Ford ES, Croft JB. Employment and activity limitations among adults with chronic obstructive pulmonary disease—United States, 2013. MMWR Morb Mortal Wkly Rep. 2015;64(11):289-295.
- Barnes PJ. Immunology of asthma and chronic obstructive pulmonary disease. Nat Rev Immunol. 2008;8(3):183-192.
- Bhat TA, Panzica L, Kalathil SG, Thanavala Y. Immune dysfunction in patients with chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2015;12(suppl 2):S169-S175.
- Agusti A, Calverley PM, Decramer M, Stockley RA, Wedzicha JA. Prevention of exacerbations in chronic obstructive pulmonary disease: knowns and unknowns. Chronic Obstr Pulm Dis. 2014;1(2):166-184.
- The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General. Rockville, MD: US Department of Health and Human Services, Public Health Service, Office of the Surgeon General. 2014. https://www.ncbi.nlm.nih.gov/books/NBK179276/pdf/Bookshelf_NBK179276.pdf. Accessed September 26, 2019.
- Adult tobacco use: glossary. Centers for Disease Control and Prevention (CDC) National Health Interview Survey. https://www.cdc.gov/nchs/nhis/tobacco/tobacco_glossary.htm. Reviewed August 29, 2017. Accessed September 26, 2019.
- Wang TW, Asman K, Gentzke AS, et al. Tobacco product use among adults—United States, 20 MMWR Morb Mortal Wkly Rep. 2018;67(44):1225-1232.
- Centers for Disease Control and Prevention (CDC). Tobacco use among middle and high school students—United States, 2000–2009. MMWR Morb Mortal Wkly Rep. 2010;59(33):1063-1068.
- Babb S, Malarcher A, Schauer G, Asman K, Jamal A. Quitting smoking among adults—United States, 2000–2015. MMWR Morb Mortal Wkly Rep. 2017;65(52):1457-1464.
- Fiore MC, Jaén CR, Baker TB, et al. Treating Tobacco Use and Dependence: 2008 Update. Rockville, MD: US Dept of Health and Human Services, Public Health Service. https://www.ncbi.nlm.nih.gov/books/NBK63952/. Published May 2008. Accessed September 26, 2019.
- Anthenelli RM, Benowitz NL, West R, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers without and without psychiatric disorders (EAGLES): a double-blind, randomized, placebo-controlled clinical trial. Lancet. 2016;387(10037):2507-2520.
- Centers for Disease Control and Prevention (CDC). The Surgeon General’s 1990 report on the health benefits of smoking cessation. Executive summary. MMWR Recomm Rep. 1990;39(RR-12):i-xv, 1-12.
- Doll R, Peto R, Boreham J, Sutherland I. Mortality in relation to smoking: 50 years’ observation on male British doctors. BMJ. 2004;328(7455):1519.
- Kochanek KD, Murphy SL, Xu J, Arias E. Deaths: final data for 2017. Natl Vital Stat Rep. 2019;68(9). https://www.cdc.gov/nchs/data/nvsr/nvsr68/nvsr68_09-508.pdf. Accessed September 26, 2019.
- Ford ES, Murphy LB, Khavjou O, Giles WH, Holt JB, Croft JB. Total and state-specific medical and absenteeism costs of COPD among adults aged ≥18 years in the United Sates for 2010 and projections through 2020. Chest. 2015;147(1):31-45.
- US Burden of Disease Collaborators. The state of US health, 1990-2016: burden of diseases, injuries, and risk factors among US states. JAMA. 2018;319(14):1444-1472.
- Barnes PJ, Kleinert S. COPD—a neglected disease. Lancet. 2004;364(9434):564-565.
- Varkey B. Chronic obstructive pulmonary disease: from nihilism to tempered optimism. Curr Opin Pulm Med. 2005;11(2):113-114.
- Chronic Obstructive Pulmonary Disease (COPD). NIH Fact Sheets. https://report.nih.gov/nihfactsheets/viewfactsheet.aspx?csid=77. Accessed September 26, 2019.
- Mannino DM. Does undiagnosed chronic obstructive pulmonary disease matter? Am J Respir Crit Care Med. 2016;194(3):250-252.
- COPD National Action Plan. National Institutes of Health. https://www.nhlbi.nih.gov/sites/default/files/media/docs/COPD%20National%20Action%20Plan%20508_0.pdf. NIH publication No. 17-HL-80 Updated February 2018. Accessed September 26, 2019.
- Mannino DM, Gagnon RC, Petty TL, Lydick E. Obstructive lung disease and low lung function in adults in the United States: data from the National Health and Nutrition Examination Survey, 1988-1994. Arch Intern Med. 2000;160(11):1683-1689.
- Lindberg A, Bjerg-Bäckland A, Rönmark E, Larsson L-G, Lundbäck B. Prevalence and underdiagnosis of COPD by disease severity and the attributable fraction of smoking: report from the Obstructive Lung Disease in Northern Sweden Studies. Respir Med. 2006;100(2):264-272.
- Martinez CH, Mannino DM, Jaimes FA, et al. Undiagnosed obstructive lung disease in the United States. Associated factors and long-term mortality. Ann Am Thorac Soc. 2015; 12(12):1788-1795.
- Ferguson GT, Enright PL, Buist AS, Higgins MW. Office spirometry for lung health assessment in adults: a consensus statement from the National Lung Health Education Program. Chest. 2000;117(4):1146-1161.
- Lee TA, Bartle B, Weiss KB. Spirometry use in clinical practice following diagnosis of COPD. Chest. 2006;129(6):1509-1515.
- Joo MJ, Lee TA, Weiss KB. Geographic variation in the use of spirometry in newly diagnosed COPD. Chest. 2008;134(1):38-45.
- Bolton CE, Ionescu AA, Edwards PH, Faulkner TA, Edwards SM, Shale DJ. Attaining a correct diagnosis of COPD in general practice. Respir Med. 2005;99(4):493-500.
- Kaminsky DA, Marcy TW, Bachand M, Irvin CG. Knowledge and use of office spirometry for the detection of chronic obstructive pulmonary disease by primary care physicians. Respir Care. 2005;50(12):1639-1648.
- Tinkelman DG, Price DB, Nordyke RJ, Halbert RJ. Misdiagnosis of COPD and asthma in primary care patients 40 years of age and over. J Asthma. 2006;43(1):75-80.
- Chapman KR, Tashkin DP, Pye DJ. Gender bias in the diagnosis of COPD. Chest. 2001;119(6):1691-1695.
- Jacobs DM, Noyes K, Zhao J, et al. Early hospital readmissions after an acute exacerbation of chronic obstructive pulmonary disease in the Nationwide Readmissions Database. Ann Am Thorac Soc. 2018;15(7):837-845.
- Goto T, Faridi MK, Gibo K, et al. Trends in 30-day readmission rates after COPD hospitalization, 2006–2012. Respir Med. 2017;130:92-97.
- Make B, Dutro MP, Paulose-Ram R, Marton JP, Mapel DW. Undertreatment of COPD: a retrospective analysis of US managed care and Medicare patients. Int J Chron Obstruct Pulmon Dis. 2012;7:1-9.
- Tinkelman D, Corsello P. Chronic obstructive pulmonary disease: the impact occurs earlier than we think. Am J Manag Care. 2003;9(11):767-771.
- Kesten S. Chronic obstructive pulmonary disease in a managed-care setting. Dis Manage Health Outcomes. 2001;9:589-599.
- Centers for Disease Control and Prevention, National Center for Health Statistics. Underlying cause of death 1999-2017 on CDC WONDER Online Database. http://wonder.cdc.gov/ucd-icd10.html. Accessed September 26, 2019.
- Ford ES, Croft JB, Mannino DM, Wheaton AG, Zhang X, Giles WH. COPD surveillance—United States, 1999–2011. Chest. 2013;144(1):284-305.
- Aryal S, Diaz-Guzman E, Mannino DM. Influence of sex on chronic obstructive pulmonary disease risk and treatment outcomes. Int J Chron Obstruct Pulmon Dis. 2014;9:1145-1154.
- Johnston LD, Miech RA, O’Malley PM, Bachman JG, Schulenberg JE, Patrick ME. Monitoring the Future National Survey Results on Drug Use 1975-2018: Overview, Key Findings on Adolescent Drug Use. Ann Arbor, MI: Institute for Social Research; 2019.
- Lundbäck B, Lindberg A, Lindström M, et al. Not 15 but 50% of smokers develop COPD?—report from the Obstructive Lung Disease in Northern Sweden Studies. Respir Med. 2003;97(2):115-122.
- Marsh S, Aldington S, Shirtcliffe P, Weatherall M, Beasley R. Smoking and COPD: what really are the risks? Eur Respir J. 2006;28(4):883-886.
- Pauwels RA, Buist AS, Calverley PMA, Jenkins CR, Hurd SS; GOLD Scientific Committee. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am J Respir Crit Care Med. 2001;163(5):1256-1276.
- Global Strategy for Asthma Management and Prevention. Global Initiative for Asthma. https://ginasthma.org/wp-content/uploads/2019/01/2018-GINA.pdf. Published 2018. Accessed September 26, 2019.
- Hurst JR, Vestbo J, Anzueto A, et al; Evaluation of COPD Longitudinally to Identify Predictive Surrogate Endpoints (ECLIPSE) Investigators. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363(12):1128-1138.
- Cooper CB. The connection between chronic obstructive pulmonary disease symptoms and hyperinflation and its impact on exercise and function. Am J Med. 2006;119(10 suppl 1):21-31.
- Sutherland ER, Cherniack RM. Management of chronic obstructive pulmonary disease. N Engl J Med. 2004;350(26):2689-2697.
- Kim V, Rogers TJ, Criner GJ. New concepts in the pathobiology of chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2008;5(4):478-485.
- Sulaiman I, Cushen B, Greene G, et al. Objective assessment of adherence to inhalers by patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2017;195(10):1333-1343.
- Hardin KA, Meyers F, Louie S. Integrating palliative care in severe chronic obstructive lung disease. COPD. 2008;5(4):207-220.
- Berge KM, Hardin KA, Coburn A, Kizziar A. Pulmonary rehabilitation: to refer or not to refer? Consultant. 2015;55(5):372-377.
- Louie S. Lies, damned lies, and asthma. Consultant. 2018;58(12):336-342.
- Frazier CA. Treating allergic asthma: some do’s and don’ts. Consultant. 1963;3(7):24-26.


