Peer Reviewed
Pneumococcal Osteomyelitis in a Vaccinated Immunocompetent Child
Authors:
Abiezer Disla, MD; Divij Pasrija, MD; Catalina Marino, MD; Talya Toledano, MD; and Yekaterina Sitnitskaya, MD
NYC Health + Hospitals/Lincoln, Bronx, New York
Citation:
Disla A, Pasrija D, Marino C, Toledano T, Sitnitskaya Y. Pneumococcal osteomyelitis in a vaccinated immunocompetent child [published online November 19, 2018]. Infectious Diseases Consultant.
A 15-month-old thriving African American girl presented after having cried all night; in the morning, she did not want to stand on her right lower extremity (RLE) or walk. There had been no history of preceding trauma, but she had had cold symptoms 1 week prior.
Physical examination. At presentation, her temperature was 38.7°C and her heart rate was 150 beats/min. In a standing position, she was not bearing weight on the RLE. There was no erythema, bruising, or swelling noted in the RLE, nor tenderness on palpation of the hip or the RLE. Range of motion in both hips and the RLE joints was normal. The review of history showed that the patient was HIV-negative, had hemoglobin type AA, and had received 3 doses of 13-valent pneumococcal conjugate vaccine (PCV13) and the Haemophilus influenzae type B vaccine.
Diagnostic tests. The white blood cell (WBC) count was elevated at 22,000/µL, with 64% neutrophils, and the C-reactive protein (CRP) level was elevated at 1.9 mg/dL. Radiographs of the right femur and hip showed no abnormalities. A blood culture was sent, and empiric treatment with vancomycin and ceftriaxone was started.
On day 2, magnetic resonance imaging (MRI) showed marrow signal changes with enhancement in the right iliac wing, consistent with acute osteomyelitis, and signs of inflammation in the adjacent muscle (Figures).
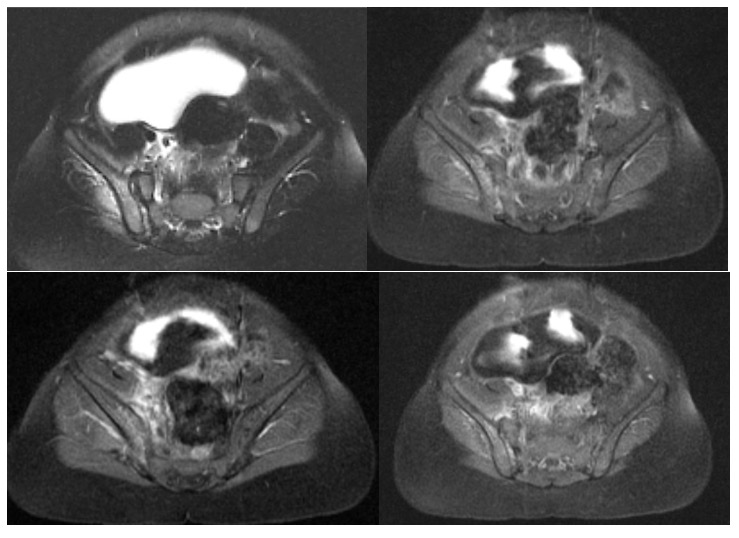
Blood culture grew pansensitive Streptococcus pneumoniae serotype 10A.
Outcome of the case. The patient’s fever resolved on day 2, and she started to walk on day 3. By day 4, the WBC count had decreased to 10,400/µL, and the CRP level had decreased to 0.9 mg/dL. Antibiotic treatment was tailored based on sensitivity and was switched to oral cephalexin on day 7. At a follow-up visit after having completed 5 weeks of antibiotic therapy, the child was asymptomatic and walking without limping.
Discussion. The Centers for Disease Control and Prevention has reported a dramatic decrease in the occurrence of invasive pneumococcal disease (IPD) among the US population after the institution of vaccination with PCV13 in 2010.1 As early as 1 year after introduction of PCV13, the incidence rate of IPD among children under 5 years decreased by almost 50%.2
Bone infection caused by S pneumoniae is rare, accounting for only 1.3% of acute osteomyelitis cases in children.2 The most common sites of osteomyelitis in children are metaphyses of long bones, most commonly of the femur, tibia, and humerus. Osteomyelitis of the pelvic bones accounts for less than 15% of all cases.3 Pelvic osteomyelitis is associated with substantial soft-tissue inflammation, particularly pyomyositis, in 85% of cases and abscesses in approximately half of cases.4 Our patient had a rare case of pelvic osteomyelitis associated with pyomyositis caused by non-PCV13 S pneumoniae serotype 10A in a vaccinated child.
Pelvic osteomyelitis and pelvic pyomyositis are difficult to diagnose clinically because of their vague presentation mimicking disease of adjacent structures, such as osteoarthritic infection of the hip.5-9 In our case, the patient presented with fever and refusal to bear weight on one lower extremity, in association with leukocytosis and elevated CRP, which raised suspicion for femoral osteomyelitis or septic arthritis of the hip. However, the patient had full range of motion in the right hip, point tenderness on palpation was not elicited anywhere, and radiographic findings of the right hip were normal. This prompted a widening of the differential diagnosis to include infection in the pelvis. Therefore, further diagnostic evaluation was employed using MRI, the most sensitive imaging modality for detecting disease in the bone and adjacent tissues.9,10
Staphylococcus aureus is the most common cause of osteoarticular infection in children, followed by Streptococcus pyogenes, while S pneumoniae is a rare cause of pediatric osteomyelitis.11-14 In our patient, bone and adjacent muscle infection in the pelvis were caused by S pneumoniae. In a published case series of pneumococcal pyomyositis, preceding trauma was found in more than a third of all patients.15 We speculate that in our patient, in the absence of trauma, an accidental seeding of pelvic bone during transient bacteremia was the primary event, which was followed by extension of infection to adjacent muscle. A total of 4 doses of PCV13 are required to complete immunization.16 Our patient was a recipient of an age-appropriate 3 doses of PCV13, but the causative S pneumoniae was nonvaccine serotype 10A. This is in concordance with recent reports of mostly nonvaccine pneumococcal serotypes causing IPD in the PCV13 era, including serotype 10A.17-19
Bone infection requires prolonged antibiotic treatment, especially when a flat bone is involved. Early diagnosis is necessary to institute appropriate treatment and prevent chronicity.12,13 In our patient’s case, treatment resulted in complete resolution of symptoms.
Thorough physical examination remains the mainstay in suspecting pelvic bone or muscle infection. Whenever a diagnosis of pelvic bone or muscle infection is a consideration, MRI is the imaging test of choice. Precise diagnosis is necessary to guide adequate treatment and prevent short- and long-term complications.
- Matanock A. Invasive pneumococcal disease in the U.S.—2008–2016. Centers for Disease Control and Prevention. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2017-10/pneumo-04-matanock.pdf. Accessed November 19, 2018.
- Krogstad P. Osteomyelitis. In: Feigin RD, Cherry JD, Demmler-Harrison GD, Kaplan SL, eds. Feigin and Cherry’s Textbook of Pediatric Infectious Diseases. Vol 1. 6th ed. Philadelphia, PA: Saunders Elsevier; 2009:725-742.
- Peltola H, Pääkkönen M. Acute osteomyelitis in children. N Engl J Med. 2014;370(4):352-360.
- Connolly SA, Connolly LP, Drubach LA, Zurakowski D, Jaramillo D. MRI for detection of abscess in acute osteomyelitis of the pelvis in children. AJR Am J Roentgenol. 2007;189(4):867-872.
- Mustafa MM, Sáez-Llorens X, McCracken GH Jr, Nelson JD. Acute hematogenous pelvic osteomyelitis in infants and children. Pediatr Infect Dis J. 1990;9(6):416-421.
- Spiegel DA, Meyer JS, Dormans JP, Flynn JM, Drummond DS. Pyomyositis in children and adolescents: report of 12 cases and review of the literature. J Pediatric Orthop. 1999;19(2):143-150.
- Kumar J, Ramachandran M, Little D, Zenios M. Pelvic osteomyelitis in children. J Pediatr Orthop B. 2010;19(1):38-41.
- Pääkkönen M, Kallio MJT, Kallio PE, Peltola H. Sensitivity of erythrocyte sedimentation rate and C-reactive protein in childhood bone and joint infections. Clin Orthop Relat Res. 2010;468(3):861-866.
- Chung T. Magnetic resonance imaging in acute osteomyelitis in children. Pediatr Infect Dis J. 2002;21(9):869-870.
- McPhee E, Eskander JP, Eskander MS, Mahan ST, Mortimer E. Imaging in pelvic osteomyelitis: support for early magnetic resonance imaging. J Pediatr Orthop. 2007;27(8):903-909.
- Branson J, Vallejo JG, Flores AR, et al. The contemporary microbiology and rates of concomitant osteomyelitis in acute septic arthritis. Pediatr Infect Dis J. 2017;36(3):267-273.
- Peltola H, Pääkkönen M, Kallio P, Kallio MJ. Short- versus long-term antimicrobial treatment for acute hematogenous osteomyelitis of childhood: prospective, randomized trial on 131 culture-positive cases. Pediatric Infect Dis J. 2010;29(12):1123-1128.
- Peltola H, Pääkkönen M, Kallio P, Kallio MJT; OM-SA Study Group. Clindamycin vs. first-generation cephalosporins for acute osteoarticular infections of childhood—a prospective quasi-randomized controlled trial. Clin Microbiol Infect. 2012;18(6):582-589.
- Olarte L, Romero J, Barson W, et al. Osteoarticular infections caused by Streptococcus pneumoniae in children in the post–pneumococcal conjugate vaccine era. Pediatr Infect Dis J. 2017;36(12):1201-1204.
- Zadroga RJ, Zylla D, Cawcutt K, et al. Pneumococcal pyomyositis: report of 2 cases and review of the literature. Clin Infect Dis. 2012;55(3):e12-e17.
- Nuorti JP, Whitney CG. Prevention of pneumococcal disease among infants and children—use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2010;59(RR-11):1-18.
- Demczuk WHB, Martin I, Griffith A, et al; Toronto Bacterial Diseases Network; Canadian Public Health Laboratory Network. Serotype distribution of invasive Streptococcus pneumoniae in Canada after the introduction of the 13-valent pneumococcal conjugate vaccine, 2010–2012. Can J Microbiol. 2013;59(12):778-788.
- Pitts SI, Apostolou A, DasGupta S, et al. Serotype 10A in case patients with invasive pneumococcal disease: a pilot study of PCR-based serotyping in New Jersey. Public Health Rep. 2015;130(1):54-519. Gaviria-Agudelo CL, Jordan-Villegas A, Garcia C, McCracken GH Jr. The effect of 13-valent pneumococcal conjugate vaccine on the serotype distribution and antibiotic resistance profiles in children with invasive pneumococcal disease. J Pediatric Infect Dis Society. 2017;6(3):253-259.


