Obesity and Cardiovascular Disease: Steps to Reduce the Risk
ABSTRACT: Obesity is associated with conditions such as hypertension, insulin resistance, dyslipidemia, and thrombosis that markedly increase the risk of cardiovascular disease and overall mortality. In addition, the pro-inflammatory and pro-thrombotic cascades triggered by the extra adipose tissue appear to increase cardiovascular risk independently. For primary and secondary prevention of cardiovascular disease, expert panels recommend that obese patients, as well as those who are overweight with comorbid conditions, lose 10% of their initial body weight. A first approach should include a program of lifestyle modification, composed of diet, exercise, and behavioral changes. Given the limited long-term efficacy of pharmacological and behavioral interventions, bariatric surgery has emerged as a prominent and durable alterative to combating obesity and its related conditions.
Key words: obesity, cardiovascular disease
_________________________________________________________________________________________________________________________________________________
A 40-year-old man presents to your office for a follow-up visit. He has a history of hypertension, obstructive sleep apnea, and gastroesophageal reflux disease.
Medications include lisinopril, hydrochlorothiazide, and omeprazole. His weight is 185 lb, height is 1.7 m, and body mass index (BMI) is 29 kg/m2. Waist circumference measures 40 inches. Blood pressure is 135/80 mm Hg. Most recent laboratory test results reveal: fasting glucose, 115 mg/dL; total cholesterol, 188 mg/dL; triglycerides, 245 mg/dL; high-density lipoprotein (HDL) cholesterol, 30 mg/dL; low-density lipoprotein (LDL) cholesterol, 110 mg/dL. He does not smoke, does not routinely exercise, and has tried several diets that he read about in magazines with no significant weight loss. He seems motivated to lose weight and asks you about weight reduction drugs.
What is this patient’s cardiovascular risk? What metabolic abnormalities are coming into play in the development of his cardiovascular risk factors? Which primary prevention interventions would you implement?
THE SCOPE OF THE PROBLEM
It does not take a trained epidemiologist to recognize that overweight and obesity have reached epidemic proportions. The scope of the problem can be easily identified by just looking around while strolling down the streets. In the United States, two thirds of the adult population is overweight or obese, 30% is considered obese, and 6% is morbidly obese.1 Rates are even higher in minority groups such as African Americans, Native Americans, and Hispanics.
More alarmingly, 30% of US children and adolescents are overweight or obese.2 A steady increase in the prevalence of childhood obesity has been documented since the 1970s, with obesity rates among adolescents nearly tripling in the past 2 decades.2 These numbers are all the more troubling for two reasons. First, childhood obesity is associated with increased prevalence of diabetes, hypertension, and dyslipidemia in the pediatric population.3 Second, it substantially raises the risk of obesity in adulthood, augmenting morbidity and mortality for future generations.4
Overweight and obesity portend serious metabolic and cardiovascular consequences, leading to insulin resistance, premature atherosclerosis, increased risk of myocardial infarction and heart failure, and decreased survival.5,6 It is estimated that the current rate of adolescent obesity will result in more than 100,000 excess cases of coronary heart disease (CHD) by 2035.7 This will have a tremendous impact on already surging overall health care costs. In 2008, the medical costs related to obesity in the United States totaled $147 billion dollars.8
Given the serious health consequences of obesity and its economic impact, this issue has gained considerable attention among medical associations and health care policy makers. The American Heart Association has issued a call for action to curb the consequences of this growing epidemic,9 which may soon overtake tobacco use as the leading cause of preventable death in the United States.10 If left unchecked, it will hamper the steady increase in life expectancy obtained in previous decades.11
DEFINING AND MEASURING OBESITY
Obesity results from an imbalance between caloric intake and expenditure. A positive caloric balance has become more prevalent in western societies as a result of sedentary lifestyles and the high caloric content of western diets. The excess energy is stored in the body in the form of adipose tissue.
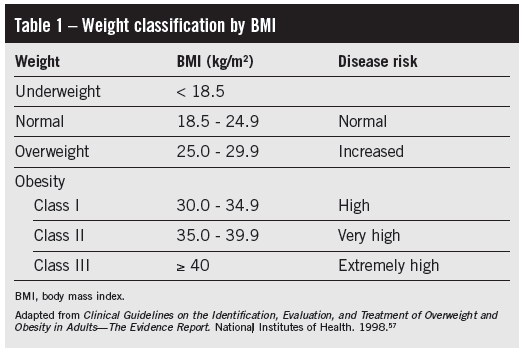
Different surrogate measures of adiposity have been developed: BMI, waist-to-hip circumference ratio (WHR), and waist circumference (WC). The latter is measured halfway between the last two ribs and the iliac crest. Of these, BMI is the most frequently used method to define the degree of obesity and quantify risk, and it is calculated from the formula: BMI = body weight (kg) / height (m2). Overweight is defined as BMI of 25 kg/m2 or higher, obesity as BMI of 30 kg/m2 or higher, and extreme (morbid) obesity as BMI of 40 kg/m2 or higher (Table 1).12 WHR and WC may be better indicators of the amount of abdominal visceral fat,13 but there is controversy about which anthropometric measure best identifies the risk of cardiovascular events. Some argue that WHR and WC are better predictors of cardiovascular risk than BMI.14 However, this notion has been challenged in an analysis of the Framingham cohort, in which there was little or no incremental value of measuring WC in addition to BMI as a predictor of coronary risk, particularly in men.15 Current guidelines recommend measurement of both BMI and WC in patients with BMI of at least 25 kg/m2, and define abdominal obesity as WC of 103 cm (40 in) or more in men, or 88 cm (35 in) or more in women.12 Additional measures such as lean body mass obtained from dual x-ray absorptiometry are used more frequently in research settings. Importantly, the latter has identified a number of patients with normal BMI but elevated body fat content (normal-weight obesity), and these patients appear to have increased cardiovascular risk.16
LINKING OBESITY WITH CARDIOVASCULAR RISK: THE METABOLIC SYNDROME
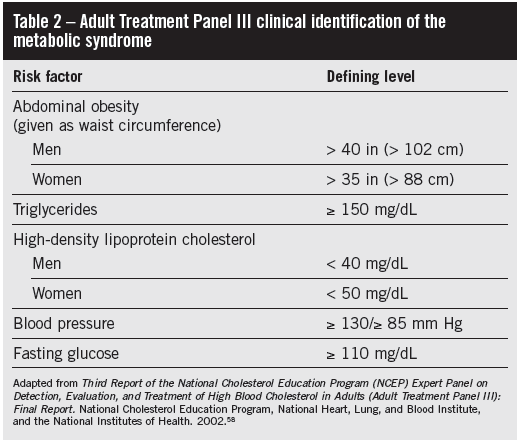
Accumulation of adipose tissue, in particular abdominal visceral fat, is associated with a cluster of metabolic conditions that appear to be ultimately mediated by insulin resistance.17 These include hypertension, atherogenic dyslipidemia, type 2 diabetes mellitus, non-alcoholic fatty liver disease, and abnormalities in coagulation. Each one of these conditions is independently associated with increased risk of cardiovascular events such as stroke or myocardial infarction. Some of these elements have been collectively labeled “metabolic syndrome,” as defined by the National Cholesterol Education Program Adult Treatment Panel III (ATP-III) (Table 2).18 Three or more of the criteria listed in the Table need to be present to fulfill the diagnosis of metabolic syndrome. In the United States, about 24% of all adults, and more than 40% of men and women over the age of 65 meet the criteria for the metabolic syndrome.19 The main purpose of this designation is the early identification of these high-risk patients, who have a 1.5- to 3-fold increased risk of CHD,20 and to respond with more aggressive strategies for prevention.
The vast majority of patients with the metabolic syndrome have abdominal obesity. Visceral fat seems to unmask the syndrome in patients with a metabolic susceptibility; the latter in turn is determined by factors such as genetics, physical activity, and drugs. It is now recognized that adipose tissue is not just a storage site for excess energy, but it is also a metabolically active tissue that produces and secretes free fatty acids (FFA) and a number of biologically active substances named adipokines.21 These factors mediate the emergence of insulin resistance that ultimately confers cardiovascular risk. FFA levels are increased in obese patients22 as a result of higher lipolitic activity of visceral fat and possibly reduced clearance.23 FFAs reach the liver directly by the portal system and interfere with the insulin receptor signaling pathways, resulting in increased hepatic glucose output. In addition, they inhibit peripheral glucose utilization by skeletal muscle, thus contributing to increased glucose levels and hyperinsulinemia.23 Chronic exposure to high FFA levels eventually contributes to beta-cell toxicity and development of type 2 diabetes mellitus.24
Triglyceride (TG) levels rise because of esterification of more available FFAs, as well as reduced lipoprotein lipase activity associated with insulin resistance. This enzyme is in charge of hydrolyzing TG in such lipoproteins as very low density lipoprotein (VLDL) and chylomicrons, and its reduced activity results in higher levels of TG-rich remnant particles (incompletely digested VLDL and chylomicrons). On the other hand, as HDL “off-loads” more TG from remnant particles, it becomes a better substrate for hepatic lipase, thereby enhancing its catabolism and reducing circulating HDL levels.25 Insulin resistance is also associated with higher number of smaller, more atherogenic LDL particles, even when the total LDL-cholesterol may be “at goal” according to current guidelines.26 Thus, measuring the number of LDL particles directly (by magnetic resonance) or indirectly (by assessing levels of apo B 100) may provide a more accurate assessment of cardiovascular risk in selected obese patients.27
It is also recognized that obesity is associated with a state of persistent, low-level inflammation, with elevated levels of pro-inflammatory cytokines such as tumor necrosis factor alpha (TNF-alpha) and interleukin (IL)-6 contributing to insulin resistance.28 A chronic state of inflammation is also associated with decreased production of nitric oxide and increased oxidative stress, leading to endothelial dysfunction and atherosclerotic plaque formation.29
In summary, visceral obesity leads to a state of chronic inflammation, insulin resistance, and atherogenic dyslipidemia, which significantly increases cardiovascular risk.
OBESITY AND THE SPECTRUM OF CARDIOVASCULAR DISEASES
Obesity has been identified as a risk factor for a wide array of cardiovascular disorders including CHD, stroke, heart failure, and arrhythmias, even after correction for other associated known risk factors such as age, hypertension, diabetes, and dyslipidemia.30
Coronary heart disease. Strong epidemiological evidence accumulated since the 1970s has identified obesity as a major, modifiable, and potentially independent risk factor for CHD.30,31 Postmortem studies in obese children and adolescents show more advanced atherosclerosis at autopsy.32 Carotid intima-medial thickness measured by ultrasound, a marker of generalized atherosclerosis and predictor of cardiovascular events, correlates directly with BMI.33 In addition, obese patients who have acute coronary syndromes tend to be younger at the time of the first cardiovascular event than their normal-weight counterparts.34
Interestingly, despite the link with obesity and premature CHD, there seems to be a paradoxical response between BMI and cardiovascular outcomes after disease manifestation. Once CHD is established, a U-shaped mortality curve by BMI has been described, whereby patients who are overweight or have mild obesity have lower mortality rates than those with normal BMI or severe obesity.35 There is controversy surrounding this potential benefit of mild obesity, also referred to as the “obesity paradox.” It has been partly attributed to bias introduced by the fact that overweight and obese patients with CHD tend to be younger, while leaner persons tend to have a higher disease burden.34 However, it may also hold true that obese patients may tolerate the stress of ischemia better than their normal-weight counterparts.
Heart failure. Obesity predisposes to heart failure by promoting hypertension, diabetes, and CHD, but an elevated BMI has also been identified as an independent risk factor.36 The rate of heart failure increases by 5% in men and 7% in women per each unit increase in BMI.36 The excessive body mass imposes stress on the heart due to compensatory hemodynamic alterations such as increased blood volume, cardiac output, and blood pressure, leading to increased filling pressures, left ventricular hypertrophy, and diastolic dysfunction. In addition, accumulation of fat tissue results in an obese-specific cardiomyopathy, named adipositas cordis,9 by two different mechanisms:
•Infiltration of the myocardium by adipose tissue, which can even lead to restrictive physiology.
•Lipotoxicity of the cardiomyocytes by substances released by epicardial fat, leading to cell death through apoptosis.
Surprisingly, once heart failure is established, it appears that overweight and obesity actually provide a survival benefit, similar to the obesity paradox described for CHD.37 The mechanisms of this protective effect are not entirely clear.
Arrhythmias. The incidence of atrial fibrillation (AF) is growing rapidly, and it is expected to soar with the aging of the population and the obesity epidemic. The risk of developing AF is almost 50% higher in obese patients compared with those who are non-obese.38 Hypertension, diastolic dysfunction, increased sympathetic tone, and sleep apnea are factors associated with both conditions and probably underlie the link between the two. Obesity also appears to increase the risk of ventricular arrhythmias and sudden cardiac death, even in the absence of cardiac dysfunction.39 Analysis of Framingham cohort data showed that obese men and women may have up to a 40-fold increase in the risk of sudden cardiac death compared with the non-obese population.39 Prolongation of the QTc interval and the presence of late potentials on signal-averaged ECG have been associated with increased BMI, potentially underlying the increased risk of ventricular arrhythmias in obese patients.40
Stroke. Numerous studies have shown an association between obesity and stroke. The risk increases by 4% for ischemic stroke and 6% for hemorrhagic stroke per each unit increase in BMI,41 even after correction for hypertension, diabetes, and cholesterol levels. The pro-thrombotic state that accompanies obesity, which is most likely secondary to increased levels of fibrinogen and plasminogen activator inhibitor-1,42 as well as the higher AF burden, likely plays a role in this association.
TREATMENT OF OBESITY AND BENEFITS OF WEIGHT LOSS
Weight control is the primary goal of therapy in obese patients for primary and secondary prevention of cardiovascular disease (CVD). Expert panels recommend that obese patients, as well as those who are overweight with comorbid conditions, lose 10% of their initial body weight.12 A first approach should include a program of lifestyle modification, composed of diet, exercise, and behavioral changes. It is clear from randomized trials that enrolling patients in such programs can achieve weight losses close to the 10% goal.43,44 Although the long-term success in sustaining these weight reductions is limited, the benefit in the associated comorbidities such as diabetes, hypertension, and dyslipidemia appears to be maintained.45,46
Dietary interventions. These alone provide only modest and non-sustained weight loss in the absence of a comprehensive lifestyle modification program. Much controversy exists about which specific type of diet achieves higher weight reductions (eg, low-fat, low-carbohydrate, Mediterranean, or low-glycemic load regimens). However, sustained caloric restriction, rather than specific macronutrient composition, is the key determinant of long-term weight control.47
Drug therapy. Weight reduction drugs can potentiate weight loss, and the best results can be expected when they are part of a comprehensive weight-management program. The National Institutes of Health/National Heart, Lung, and Blood Institute guidelines recommend that pharmacotherapy be considered for those patients who have a BMI of 30 kg/m2 or higher, or 27 kg/m2 or higher with at least one serious comorbidity.12 Unfortunately, weight is quickly regained in most cases once the agent is stopped. In addition, these medications are fraught with side effects that limit their use. Many of these drugs are appetite suppressants that act on the hypothalamus. They share amphetamine-like properties, which can increase blood pressure and trigger arrhythmias—and therefore increase cardiovascular risk. Agents such as fenfluramine and sibutramine, initially approved by the FDA, were later withdrawn from the market because of increased risk of valvular disease with the former, and higher rates of myocardial infarction and stroke with the latter. Until recently, the only centrally active appetite suppressants approved by the FDA for short-term use were phentermine and diethylpropion; long-term studies with these agents are unavailable. In 2012, the FDA approved two new agents for weight reduction: lorcaserin, a selective serotonin receptor agonist, and the combination of phentermine and topiramate. Both drugs had previously been denied FDA approval in 2010 citing safety concerns. However, the introduction of new data in 2012 led to the agency’s endorsement.
Orlistat, a peripherally acting agent that blocks intestinal fat absorption by inhibiting pancreatic and gastric lipase, is also FDA approved for long-term use. Its effects are relatively mild, achieving less than 5% weight loss compared to placebo. However, it did reduce the incidence of diabetes in patients with preexisting impaired glucose tolerance after 4-year follow-up.48 Most common side effects are gastrointestinal tract–related. Orlistat can also inhibit the absorption of lipophilic vitamins and medications.
Bariatric surgery. Given the limited long-term efficacy of pharmacological and behavioral interventions, bariatric surgery has emerged as a prominent and durable alterative to combating obesity and its related conditions. In the United States, the number of bariatric surgical procedures performed annually has grown exponentially in the last 15 years. Increasing expertise in the surgical community has resulted in perioperative morbidity and mortality comparable to that of other major operations. The 30-day risk of death after bariatric surgery is reported at 0.3%, and the rate of major complications at 4.1%.49 It is important to note, however, that these numbers correspond to high-volume bariatric centers, and may not be more widely attainable.
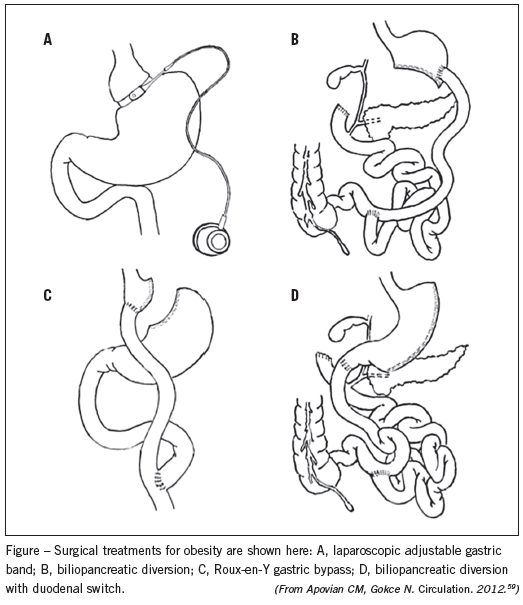
Bariatric surgery encompasses several different procedures: Roux-en-Y gastric bypass, laparoscopic adjustable gastric band, and biliopancreatic diversion with or without duodenal switch (Figure). The Roux-en-Y gastric bypass (laparoscopic or open approaches) and the laparoscopic adjustable gastric band account for more than 90% of the bariatric procedures performed worldwide.50 Current guidelines recommend these operations for patients with a BMI of 40 kg/m2 or higher, or 35 kg/m2 or higher with at least one serious comorbidity.12 The long-term benefits of these procedures were demonstrated in the Swedish Obese Subjects study, with continued weight loss 10 years after surgery ranging between 14% and 25% from baseline weight depending on the specific type of procedure performed. This translated into continued recovery from diabetes, hypertension, dyslipidemia, and sleep apnea more than 10 years after surgery, and an impressive 23% reduction in mortality compared with control subjects.51
Similarly, in a retrospective study involving almost 8000 patients who underwent bariatric surgery, long-term mortality was reduced by 40% compared with matched controls.52 Most recently, two small randomized trials showed that bariatric surgery was superior to medical therapy as a treatment strategy for diabetes in obese patients.53,54 In one of these studies, the rate of complete remission from diabetes at 2 years was 75% for gastric bypass and 95% for biliopancreatic diversion, compared with no remission for medical therapy.53 This kind of success will likely lead to the inclusion of bariatric surgery in the treatment algorithms of diabetes, as well as testing surgical options in less severely obese patients. Indeed, one type of laparoscopic adjustable gastric band was recently approved by the FDA for a lower BMI indication (BMI of 30 to 40 kg/m2 with at least one serious comorbidity).
Unfortunately, the success of bariatric surgery is also a reflection of our failure to contain the growing epidemic of obesity and its related comorbidities with primary prevention strategies. Although in practiced hands morbidity is low, bariatric surgery is not entirely without risk in the perioperative period. There are also long-term complications including micronutrient deficiencies (particularly vitamin B12, folate, and iron), gastrointestinal symptoms, osteoporosis, and major depression.
CASE STUDY DISCUSSION AND CONCLUDING REMARKS
Our patient should be labeled as overweight according to his BMI, and he meets the definition for abdominal obesity according to his WC. His 10-year risk of CVD according to the Framingham CVD risk score is 9%,55 which can be labeled as intermediate. However, he meets 5 out of the 5 criteria used for the diagnosis of metabolic syndrome, and this should alert the physician that this patient’s risk may be actually higher. Thus, he may benefit from more aggressive strategies for prevention. Newer prediction models such as the Reynolds score, which incorporates additional variables including family history, hemoglobin A1c, and C-reactive protein levels, appear to have more discriminative power than the Framingham score, especially in women.56 Our patient should be enrolled in an intensive lifestyle modification program, with emphasis on diet, exercise and behavioral changes, and with the goal to obtain a 10% weight reduction. This will help control his blood pressure and insulin resistance. Although his LDL cholesterol is at goal according to the ATP-III guidelines, the high non-HDL cholesterol and the ratio of TG/HDL-C of more than 3 are indicative of higher risk. Pharmacological interventions for primary prevention such as low-dose aspirin, as well as niacin for hypertriglyceridemia, may be considered. Weight reduction drugs are not indicated without concomitant lifestyle modifications, are not likely to result in sustained weight loss, and are fraught with side effects, so he is unlikely to benefit from them. According to current guidelines, our patient is not a candidate for bariatric surgery.
In summary, obesity is a pathological metabolic state associated with conditions such as hypertension, insulin resistance, dyslipidemia, and thrombosis that markedly increase the risk of CVD and overall mortality. In addition, the pro-inflammatory and pro-thrombotic cascades triggered by the extra adipose tissue appear to increase cardiovascular risk independently of the aforementioned comorbidities. The growing prevalence of obesity has become a critical problem worldwide. Further public health initiatives need to be developed and implemented for the prevention, early detection, and treatment of obesity.
1. Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999 2010. JAMA.2012;307(5):491-497.
2. Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity and trends in body mass index among US children and adolescents, 1999-2010. JAMA.2012;307(5):483-490.
3. Freedman DS, Dietz WH, Srinivasan SR, Berenson GS. The relation of overweight to cardiovascular risk factors among children and adolescents:
the Bogalusa Heart Study. Pediatrics. 1999;103(6 Pt 1):1175-1182.
4. Steinberger J, Daniels SR. Obesity, insulin resis-tance, diabetes, and cardiovascular risk in children: an American Heart Association scientific statement from the Atherosclerosis, Hypertension, and Obesity in the Young Committee (Council on Cardiovascular Disease in the Young) and the Diabetes Committee (Council on Nutrition, Physical Activity, and Metabolism). Circulation. 18 2003;107(10):1448-1453.
5. Berrington de Gonzalez A, Hartge P, Cerhan JR, et al. Body-mass index and mortality among 1.46 million white adults. N Engl J Med. 2010;363(23):
2211-2219.
6. Whitlock G, Lewington S, Sherliker P, et al. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet. 2009;373(9669):1083-1096.
7. Bibbins-Domingo K, Coxson P, Pletcher MJ, Lightwood J, Goldman L. Adolescent overweight and future adult coronary heart disease. N Engl J Med. 2007;357(23):2371-2379.
8. Finkelstein EA, Trogdon JG, Cohen JW, Dietz W. Annual medical spending attributable to obesity: payer-and service-specific estimates. Health Aff (Millwood).2009;28(5):w822-831.
9. Poirier P, Giles TD, Bray GA, et al. Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss: an update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism. Circulation. 2006;113(6):898-918.
10. Lavie CJ, Milani RV, Ventura HO. Obesity and cardiovascular disease: risk factor, paradox, and impact of weight loss. J Am Coll Cardiol. 2009;53(21):1925-1932.
11. Ford ES, Capewell S. Coronary heart disease mortality among young adults in the U.S. from 1980 through 2002: concealed leveling of mortality rates. J Am Coll Cardiol. 2007;50(22):2128-2132.
12. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults—The Evidence Report. National Institutes of Health. Obes Res. 1998;6(Suppl 2):51S-209S.
13. Pouliot MC, Despres JP, Lemieux S, et al. Waist circumference and abdominal sagittal diameter: best simple anthropometric indexes of abdominal visceral adipose tissue accumulation and related cardiovascular risk in men and women. Am J Cardiol. 1994;73(7):460-468.
14. Yusuf S, Hawken S, Ounpuu S, et al. Obesity and the risk of myocardial infarction in 27,000 participants from 52 countries: a case-control study. Lancet.2005;366(9497):1640-1649.
15. Freiberg MS, Pencina MJ, D’Agostino RB, et al. BMI vs. waist circumference for identifying vascular risk. Obesity (Silver Spring). 2008;16(2):463-469.
16. Kosmala W, Jedrzejuk D, Derzhko R, et al. Left ventricular function impairment in patients with normal-weight obesity: contribution of abdominal fat deposition, profibrotic state, reduced insulin sensitivity, and proinflammatory activation. Circ Cardiovasc Imaging. 2012;5(3):349-356.
17. Reaven GM. Insulin resistance, the insulin resistance syndrome, and cardiovascular disease. Panminerva Med. 2005;47(4):201-210.
18. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002;106(25):3143-3421.
19. Ford ES, Giles WH, Dietz WH. Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey.JAMA. 2002;287(3):356-359.
20. Isomaa B, Almgren P, Tuomi T, et al. Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care. 2001;24(4):
683-689.
21. Deng Y, Scherer PE. Adipokines as novel biomarkers and regulators of the metabolic syndrome. Ann N Y Acad Sci. 2010;1212:E1-E19.
22. Opie LH, Walfish PG. Plasma free fatty acid concentrations in obesity. N Engl J Med. 1963;268:757-760.
23. Boden G. Obesity, insulin resistance and free fatty acids. Curr Opin Endocrinol Diabetes Obes. 2011;18(2):139-143.
24. Giacca A, Xiao C, Oprescu AI, Carpentier AC, Lewis GF. Lipid-induced pancreatic beta-cell dysfunction: focus on in vivo studies. Am J Physiol Endocrinol Metab. 2011;300(2):E255-262.
25. Lewis GF, Rader DJ. New insights into the regulation of HDL metabolism and reverse cholesterol transport. Circ Res. 2005;96(12):1221-1232.
26. Dayspring TD, Pokrywka G. Impact of triglycerides on lipid and lipoprotein biology in women. Gend Med. 2010;7(3):189-205.
27. Davidson MH, Ballantyne CM, Jacobson TA, et al. Clinical utility of inflammatory markers and advanced lipoprotein testing: advice from an expert panel of lipid specialists. J Clin Lipidol. 2011;5(5):338-367.
28. Fernandez-Real JM, Ricart W. Insulin resistance and chronic cardiovascular inflammatory syndrome. Endocr Rev. 2003;24(3):278-301.
29. Virdis A, Santini F, Colucci R, et al. Vascular generation of tumor necrosis factor-alpha reduces nitric oxide availability in small arteries from visceral fat of obese patients. J Am Coll Cardiol. 2011;58(3):238-247.
30. Hubert HB, Feinleib M, McNamara PM, Castelli WP. Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham Heart Study. Circulation. 1983;67(5):968-977.
31. Manson JE, Colditz GA, Stampfer MJ, et al. A prospective study of obesity and risk of coronary heart disease in women. N Engl J Med. 1990;322(13):882-889.
32. Strong JP, Malcom GT, McMahan CA, et al. Prevalence and extent of atherosclerosis in adolescents and young adults: implications for prevention from the Pathobiological Determinants of Atherosclerosis in Youth Study. JAMA. 1999 281(8):727-735.
33. Freedman DS, Dietz WH, Tang R, et al. The relation of obesity throughout life to carotid intima-media thickness in adulthood: the Bogalusa Heart Study. Int J Obes Relat Metab Disord. 2004;28(1):159-166.
34. Eisenstein EL, Shaw LK, Nelson CL, et al. Obesity and long-term clinical and economic outcomes in coronary artery disease patients. Obes Res. 2002;10(2):83-91.
35. Romero-Corral A, Montori VM, Somers VK, et al. Association of bodyweight with total mortality and with cardiovascular events in coronary artery disease: a systematic review of cohort studies. Lancet. 2006;368(9536):666-678.
36. Kenchaiah S, Evans JC, Levy D, et al. Obesity and the risk of heart failure. N Engl J Med. 2002;347(5):305-313.
37. Horwich TB, Fonarow GC, Hamilton MA, et al. The relationship between obesity and mortality in patients with heart failure. J Am Coll Cardiol. 2001;38(3):789-795.
38. Wanahita N, Messerli FH, Bangalore S, et al. Atrial fibrillation and obesity—results of a meta-analysis. Am Heart J. 2008;155(2):310-315.
39. Kannel WB, Plehn JF, Cupples LA. Cardiac failure and sudden death in the Framingham Study. Am Heart J. 1988;115(4):869-875.
40. el-Gamal A, Gallagher D, Nawras A, et al. Effects of obesity on QT, RR, and QTc intervals. Am J Cardiol. 1995;75(14):956-959.
41. Kurth T, Gaziano JM, Berger K, et al. Body mass index and the risk of stroke in men. Arch Intern Med. 2002;162(22):2557-2562.
42. Kaye SM, Pietilainen KH, Kotronen A, et al. Obesity-related derangements of coagulation and fibrinolysis: a study of obesity-discordant monozygotic twin pairs. Obesity (Silver Spring). 2012;20(1):88-94.
43. Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med.2002;346(6):393-403.
44. Pi-Sunyer X, Blackburn G, Brancati FL, et al. Reduction in weight and cardiovascular disease risk factors in individuals with type 2 diabetes: one-year results of the look AHEAD trial. Diabetes Care. 2007;30(6):1374-1383.
45. Knowler WC, Fowler SE, Hamman RF, et al. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet. 2009;374(9702):1677-1686.
46. Wing RR. Long-term effects of a lifestyle inter-vention on weight and cardiovascular risk factors in individuals with type 2 diabetes mellitus: four-year results of the Look AHEAD trial. Arch Intern Med. 2010;170(17):1566-1575.
47. Wadden TA, Webb VL, Moran CH, Bailer BA. Lifestyle modification for obesity: new developments in diet, physical activity, and behavior therapy. Circulation.2012;125(9):1157-1170.
48. Torgerson JS, Hauptman J, Boldrin MN, Sjostrom L. XENical in the prevention of diabetes in obese subjects (XENDOS) study: a randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care. 2004;27(1):155-161.
49. Flum DR, Belle SH, King WC, et al. Perioperative safety in the longitudinal assessment of bariatric surgery. N Engl J Med. 2009;361(5):445-454.
50. Robinson MK. Surgical treatment of obesity—weighing the facts. N Engl J Med. 2009;361(5):520-521.
51. Sjostrom L, Narbro K, Sjostrom CD, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357(8):741-752.
52. Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357(8):753-761.
53. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366(17):1577-1585.
54. Schauer PR, Kashyap SR, Wolski K, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med. 2012;366(17):1567-1576.
55. D’Agostino RB Sr, Vasan RS, Pencina MJ, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation. 2008;117(6):743-753.
56. Cook NR, Paynter NP, Eaton CB, et al. Comparison of the Framingham and Reynolds Risk scores for global cardiovascular risk prediction in the multiethnic Women’s Health Initiative. Circulation. 2012;125(14):1748-1756, S1741-1711.
57. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults—The Evidence Report. National Institutes of Health. National Heart, Lung and Blood Institute. Bethesda (MD): National Heart, Lung, and Blood Institute and the National Institute of Diabetes and Digestive and Kidney Diseases; 1998:xvii. NIH publication no. 98-4083.
58. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): Final Report. Bethesda (MD): National Cholesterol Education Program, National Heart, Lung, and Blood Institute, and the National Institutes of Health; 2002:II-27. NIH publication no. 02-5215.
59. Apovian CM, Gokce N. Obesity and cardiovascular disease. Circulation. 2012;125(9):1178-1182.


