Peer Reviewed
A Young Man With E-Cigarette or Vaping-Associated Lung Injury
AUTHORS:
Omar Elmasry, MD1 • Sung Y. Chae, MD2,3 • James Ding, BA2
AFFILIATIONS:
1PGY-3 Resident, JFK Family Medicine Residency Program, Edison, New Jersey
2Assistant Director, JFK Family Medicine Residency Program, Edison, New Jersey
3Hackensack Meridian School of Medicine, Nutley, New Jersey
CITATION:
Chae SY, Ding J, Elmasry O. A young man with e-cigarette or vaping-associated lung injury. Consultant. 2022;62(1):e29-e32. doi:10.25270/con.2021.04.00015
Received December 8, 2020. Accepted January 13, 2021. Published online April 29, 2021.
DISCLOSURES:
The authors report no relevant financial relationships.
CORRESPONDENCE:
Sung Y. Chae, MD, JFK Family Medicine Residency Program, 65 James Street, Edison, NJ 08820 (sung.chae@hmhn.org)
A 22-year-old man presented to our emergency department (ED) with a 6-day history of intermittent fever, shortness of breath, cough, diarrhea, and chills. He had gone to a bar with friends who were visiting from out of state about 1 week prior to presentation. Two days prior to presentation, he had gone to an urgent care facility, where he received a diagnosis of pneumonia and was prescribed azithromycin and an albuterol inhaler. Lack of improvement and increasing exertional dyspnea prompted him to present to our ED. He also noted nausea and a weight loss of 10 lbs during this period.
History. The patient had asthma as a child, but it had not been active recently. He had a prior hospitalization for sepsis caused by a leg abscess in 2013 but had been otherwise healthy. He said he had been vaping nicotine and marijuana for about a year.
Physical examination. Upon admission to our ED, the patient’s temperature was 37.3 °C, pulse was 115 bpm, respiratory rate was 36 br/min, and pulse oximetry was 82% on room air, which improved to 96% when a non-rebreather mask was used.
He appeared ill, mildly diaphoretic, and had conversational dyspnea. He had tachycardia, but no murmurs were observed. His breath sounds were decreased bilaterally with no wheezing or rales. Abdominal examination findings were unremarkable. There was no tenderness or edema of his lower extremities.
Diagnostic testing. Results of laboratory tests were remarkable for leukocytosis of 18.9 with neutrophilia, a ferritin level of 960 ng/mL (reference range, 20-250 ng/mL), a D-dimer level of 3.03 (normal, < 0.50), a C-reactive protein level of 37.3 (normal, < 10 mg/L), an aspartate aminotransferase level of 76 U/L (reference range, 8-48 U/L), and an alanine aminotransferase level of 69 U/L (reference range, 7-55 U/L).
A respiratory viral panel was negative for multiple pathogens, including non-SARS-CoV-2 coronaviruses, influenza A and B, and parainfluenza, as well as for pertussis, Mycoplasma and Chlamydophila pneumoniae by polymerase chain reaction (PCR) test. Streptococcus pneumoniae and Legionella urine antigens were negative.
A chest radiography scan was conducted, results of which showed patchy air space opacities (Figure 1). A computed tomography (CT) angiogram of the chest showed no pulmonary embolus but did show bilateral, diffuse ground-glass opacities (Figure 2). Results of a urine drug screen were positive for tetrahydrocannabinol (THC). SARS-CoV-2 RNA NAAT run on cepheid PCR was negative.
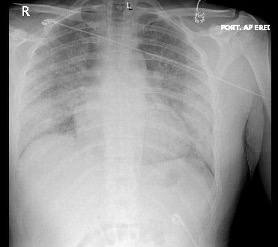

Discussion. A clinical diagnosis of e-cigarette or vaping-associated lung injury (EVALI) was made after COVID-19 was ruled out. EVALI is an uncommon, acute complication linked to the use of e-cigarettes and vaping, particularly with the use of THC, that most commonly presents in young adults with a median age of 22 to 24 years.1 As of February 2020, the Centers for Disease Control and Prevention (CDC) reported 2807 hospitalizations and 68 deaths caused by EVALI.2 However, the symptoms and ancillary findings of EVALI are similar to COVID-19, making diagnostic distinction challenging (Table).3-5 Systemic inflammation underlies both illnesses, and they can both present with acute respiratory and/or gastrointestinal symptoms and systemic signs such as fever, chills, tachycardia, and weight loss.1,3,4
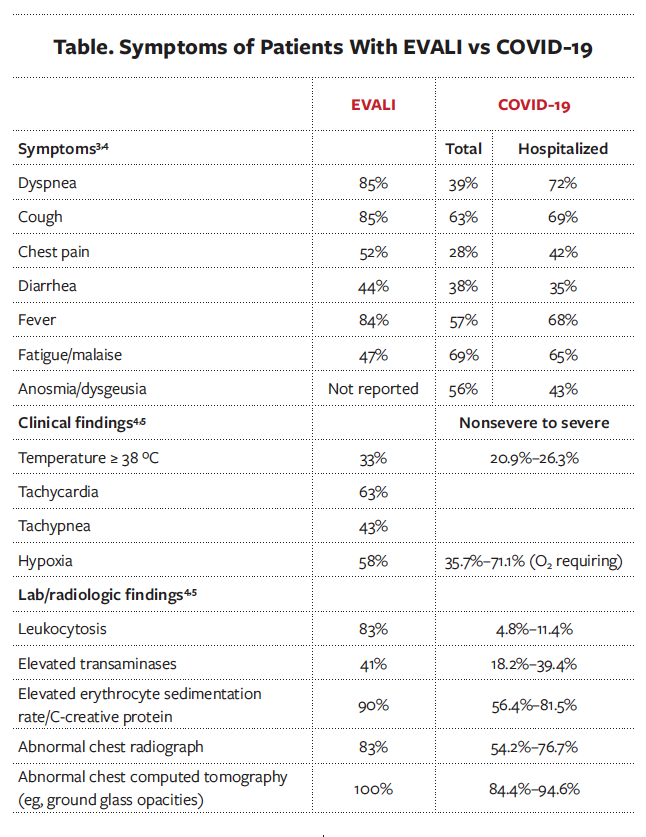
This patient’s clinical presentation, imaging, and laboratory results were consistent with acute pneumonia. Thus, bacterial or viral infection needed to be ruled out, and empiric antibiotics were appropriate. Common respiratory pathogens—including Streptococcus pneumoniae, Mycoplasma pneumoniae, Legionella pneumophila, and influenza A and B during flu season— should be ruled out.1 In addition, given the high level of inflammatory markers, hypoxia, and elevated D-dimer level, pulmonary embolism had to be ruled out as well, which was achieved via the CT angiogram of the chest. This CT showed diffuse ground glass opacities, which are found in patients with both COVID-19 and EVALI. However, there was subpleural sparing (Figure 2), which is more consistent with EVALI.6 Furthermore, nausea and weight loss are commonly associated with EVALI.4
Another distinguishing factor is that patients with COVID-19 present at a median age of 43 to 47 years, whereas EVALI presents in much younger individuals in their 20s, predominately men. However, this distinction is not absolute, and as with COVID-19, EVALI patients older than 50 years of age and those with underlying cardiac and pulmonary comorbidities have a higher risk for needing critical care, including mechanical ventilation and prolonged hospitalization. As a result, vaping and e-cigarette exposure should be considered in older populations. Empiric treatment, particularly with corticosteroids, may be lifesaving in these circumstances.1 There is concern that both cigarette and e-cigarette/vaping elevate the expression of angiotensin-converting enzyme 2 in the lungs, thereby increasing susceptibility to COVID-19. If present together, these entities can potentially worsen the inflammatory response and cause a “double-hit” situation that needs to be considered in those with COVID-19 and an e-cigarette/vaping history.7
Vitamin E acetate, an additive to illicit THC-containing products, has been implicated as a potential causative agent in EVALI. In a recent study, vitamin E acetate was found in 94% of bronchoalveolar lavage specimens of 51 patients with EVALI compared with none among 99 controls who were nonsmokers (n = 52), cigarette smokers (n = 29), or exclusive users of nicotine e-cigarettes (n = 18).8 Inhaled vitamin E acetate appears to generate an inflammatory response, disrupting the surfactant layer and causing lung dysfunction.7 However, this is not definitive, and given the many potential pulmonary toxins in e-cigarettes and vaping products (whether THC- or nicotine-based), more research is required to elucidate the underlying mechanism of lung injury.4,8
Treatment and management. Patients should be hospitalized if they exhibit hypoxia and any signs of respiratory distress. Corticosteroids have shown efficacy in improving outcomes with both EVALI1,4 and COVID-19.9,10 However, steroids are often given in combination with empiric antibiotics and antivirals (particularly during flu season), as EVALI is a diagnosis of exclusion and there are no tests to clearly rule in this condition. Corticosteroids likely dampen the inflammatory response in these entities, since both lead to significant inflammatory lung damage. Among 140 patients with EVALI treated with corticosteroids who were reported to the CDC, 82% were noted to have improved.1 As a result, unless there is a clear contraindication, such as a strong suspicion for fungal pneumonia, empiric treatment with steroids should strongly be considered.
Once discharged from the hospital, follow-up within 1 to 2 weeks is recommended with repeat pulse oximetry and chest radiography scans. The potential long-term impacts of EVALI are unknown, and it is strongly recommended that these patients receive the influenza vaccine and consider receiving the pneumococcal vaccine as well. Smoking/vaping cessation is strongly advised, as continued use may lead to recurrence of symptoms and further lung injury.1
Patient outcome. Our patient was admitted to the hospital and received intravenous (IV) hydration, as well as 1 dose of IV azithromycin and doxycycline, prior to being switched to IV ceftriaxone and levofloxacin by the infectious diseases specialist. Dexamethasone, 6 mg, IV twice daily was added for a total of 5 days. A pulmonologist consultation resulted in the patient being transferred to the intensive care unit and switched to high-flow nasal cannula at 50 L/min, with a fraction of inspired oxygen of 100%. Two inpatient COVID-19 tests were conducted, results of which were negative. An outpatient COVID-19 test from his urgent care visit had later returned negative results as well (delays in receiving results from the overwhelmed laboratory were noted).
The patient responded well to treatment and was gradually weaned off oxygen by hospital day 7, with improvement in his white blood cell count, transaminase level, and inflammatory marker levels. He was counseled on smoking/vaping cessation, discharged the following day on oral levofloxacin, and advised to follow up with his primary care physician and pulmonary specialist.
References
1. Siegel DA, Jatlaoui TC, Koumans EH, et al; Lung Injury Response Clinical Working Group and Lung Injury Response Epidemiology/Surveillance Group. Update: interim guidance for health care providers evaluating and caring for patients with suspected e-cigarette, or vaping, product use associated lung injury-United States, October 2019. MMWR Morb Mortal Wkly Rep. 2019;68(41):919-927. https://doi.org/10.15585/mmwr.mm6841e3
2. Centers for Disease Control and Prevention. Outbreak of lung injury associated with the use of e-cigarette, or vaping, products. Reviewed November 27, 2020. Accessed July 28, 2020. https://www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease.html#overview
3. Tenforde MW, Billing Rose E, Lindsell CJ, et al; CDC COVID-19 Response Team. Characteristics of adult outpatients and inpatients with COVID-19 – 11 academic medical centers, United States, March-May 2020. MMWR Morb Mortal Wkly Rep. 2020;69(26):841-846. http://dx.doi.org/10.15585/mmwr.mm6926e3
4. Layden JE, Ghinai I, Pray I, et al. Pulmonary illness related to e-cigarette use in Illinois and Wisconsin – final report. N Engl J Med. 2020;382(10):903-916. https://doi.org/10.1056/NEJMoa1911614
5. Guan W, Ni Z, Hu Y, et al; China Medical Treatment Expert Group for COVID-19. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708-1720. https://doi.org/10.1056/NEJMoa2002032
6. Ansari-Gilani K, Petraszko A, Gilkeson RC. COVID-19 pneumonia versus EVALI, distinguishing the overlapping CT features in the COVID-19 era. Heart Lung. 2020;49(6):P885-P886. https://doi.org/10.1016/j.hrtlng.2020.06.008
7. Pino LE, Triana I, Pérez C, et al. Electronic nicotine delivery systems (ECs) and COVID-19: the perfect storm for young consumers. Clin Translat Onc. 2021;23:5-9. https://doi.org/10.1007/s12094-020-02391-x
8. Blount BC, Karwowski MP, Shields PG, et al. Vitamin E acetate in brochoalveolar-lavage fluid associated with EVALI. N Engl J Med. 2020;382(8):697-705. https://doi.org/10.1056/NEJMoa1916433
9. National Institutes of Health. COVID-19 treatment guidelines. Accessed September 8, 2020. https://www.covid19treatmentguidelines.nih.gov/immunomodulators/corticosteroids
10. World Health Organization. Corticosteroids for COVID-19. Published September 2, 2020. Accessed September 7, 2020. https://www.who.int/publications/i/item/WHO-2019-nCoV-Corticosteroids-2020


