Peer Reviewed
Postoperative Naloxone-Induced Pulmonary Edema
AUTHORS:
Hunaid Adam Gurji, DO, PhD1,2 • Angelito Andres Ham, MD3,4
AFFILIATIONS:
1Department of Pediatrics, The University of Texas Health Science Center at Houston, Houston, Texas
2 Department of Pediatrics, Memorial Hermann Sugar Land Hospital, Sugar Land, Texas
3 US Anesthesia Partners, Houston, Texas
2Department of Anesthesia, Memorial Hermann Sugar Land Hospital, Sugar Land, Texas
CITATION:
Gurji HA, Ham AA. Postoperative pulmonary edema in an adolescent. Consultant. 2021;61(10):e33-e36. doi:10.25270/con.2021.02.00010
Received August 16, 2020. Accepted November 20, 2020. Published online February 19, 2021.
DISCLOSURES:
The authors report no relevant financial relationships.
CORRESPONDENCE:
Hunaid Adam Gurji, DO, PhD, University of Texas Health Science Center at Houston, Department of Pediatrics, 17500 W. Grand Parkway South, Sugar Land, TX, 77479 (hunaid.a.gurji@uth.tmc.edu)
A 13-year-old boy presented to our pediatric emergency department (ED) with acute onset of right testicular pain, which had started approximately 4 hours prior to arrival.
Physical examination. The patient was well-appearing except for the testicular pain. He weighed 60 kg and did not report any allergies. He did not have any significant medical history. The right side of his scrotum was noted to be swollen and tender. At the time of presentation, the patient’s temperature was 36.5 °C, heart rate was 78 bpm, oxygen saturation was 100% on room air, and his blood pressure was 132/81 mmHg.
Diagnostic tests. In the ED, an ultrasonography scan with doppler was performed on the scrotum and testes. Results showed that there was no blood flow to the right testicle. The patient underwent surgery for detorsion of the right teste with bilateral orchiopexy and excision of bilateral appendix epididymis. Postoperatively, he was taken to the post-anesthesia care unit, where he experienced hypoventilation with a low-oxygen saturation. A jaw thrust was administered, and ventilation was assisted with a bag-mask. His oxygen saturation increased to 99%.
Because he still required ventilatory assistance, it was suspected that the patient had narcotic-induced respiratory depression. Naloxone was given in divided doses to a total dose of 200 mcg. He started to become arousable and followed commands. Shortly thereafter, he began coughing and subsequently coughed up pink frothy sputum. His oxygen level desaturated, and he was placed on a nonrebreather facemask.
A chest radiography scan showed concern for pulmonary edema (Figure 1). He was placed on bilevel positive airway pressure (BiPAP) at 14/7 cmH20. After another cough with blood-tinged sputum, he was reintubated to protect his airway given the concern for pulmonary hemorrhage. The patient’s average heart rate and blood pressure pre- and post-naloxone administration were 78 bpm and 157/88 mmHg and 83 bpm and 119/75 mmHg, respectively.

Figure 1. Development of bilateral pulmonary edema after administration of naloxone (total 200 mcg).
Differential diagnosis. The differential diagnosis for postoperative hemoptysis is broad. Diagnosis could include naloxone-induced negative pressure pulmonary edema, naloxone-induced pulmonary edema, traumatic intubation, laceration of the lips during intubation, or gastrointestinal bleed. Other differentials, which are less likely in this scenario, include epistaxis, tuberculosis exposure, or pulmonary infection. According to documentation, the initial intubation for his surgery was uneventful. Moreover, no lip or tongue lacerations were noted after reintubation. Thus, traumatic intubation and lip lacerations were unlikely. Since his vital signs were normal prior to surgery and he was afebrile, infectious etiology was also unlikely. No tubes or devices were inserted into the patient's stomach prior to the development of blood-tinged sputum. Therefore, gastrointestinal bleeding was unlikely as well.
After the patient was reintubated, he was placed on a ventilator with a positive end-expiratory pressure (PEEP) of 8 cmH20. However, after blood appeared in the endotracheal tube, the PEEP was subsequently increased to 10 cmH20, because of the concern for pulmonary hemorrhage. The patient was transferred to the pediatric intensive care unit. A nasogastric tube was placed, and the output appeared to be pink frothy fluid. A venous blood gas demonstrated respiratory acidosis. Thus, a pediatric pulmonologist was consulted.
Findings from a bronchoscopy demonstrated thick sticky blood-tinged secretions in the posterior wall of the trachea at the tip of the endotracheal tube, but no active bleeding was seen. His anatomy was average for an adolescent boy. Mucosal inflammation of an unclear etiology was present but was likely secondary to the direct effect of blood. Naloxone-induced pulmonary edema was ultimately diagnosed, since the majority the patient’s symptoms appeared to occur after the naloxone administration.
Discussion. Naloxone is a medication used to reverse opioid overdose. It competitively blocks opioids at the mu, kappa, and delta opioid receptors. Although this medication is lifesaving, many unexpected adverse effects can develop. These include, but are not limited to, hypertension, tachycardia, agitation, dizziness, diaphoresis, or abdominal cramps.1 One rare adverse event is the development of pulmonary edema.2–5 Therefore, naloxone should be used with caution in patients with pre-existing heart conditions. One study4 described how a patient who underwent cardiac bypass had developed pulmonary edema after receiving 0.4 mg of naloxone. However, patients without cardiac disease can also develop this issue.6
Naloxone can lead to laryngospasm, which is a type of upper airway obstruction. During reversal of opioid use or anesthesia, a patient may inhale against a closed glottis (the Müeller maneuver) and develop an increase in negative intrapulmonary pressure. This allows for increased venous return to the right side of the heart, which leads to an increase in pulmonary vascular volume and pressure. Because of the pressures generated with this upper airway obstruction, a pressure gradient is generated that favors fluid drawn into the alveolar spaces inhibiting gaseous exchange.7-10 This post-obstructive negative pressure pulmonary edema formation has been shown to develop among 0.05% to 0.1% in those undergoing intubation and general anesthesia.11
In addition, naloxone can cause pulmonary edema.5,12 Naloxone competitively blocks opioids from binding to their receptors in the central nervous system, which in the presence of painful stimuli, allows for reactivation of the central pain pathways leading to a surge of catecholamines.13-16
This catecholaminergic surge leads to a sympathetic response, which usually manifests as tachycardia or hypertension. One review17 described cases where the neurogenic release of these catecholamines redirects systemic blood to the pulmonary circuit. Additionally, the surge in catecholamines can cause pulmonary vasoconstriction, pulmonary hypertension, and increased permeability of the pulmonary capillaries.18 The surge in pulmonary blood volume and pressure can lead to pulmonary edema or neurogenic pulmonary edema. A case report concluded that naloxone administration-induced catecholamine release can lead to development of pulmonary edema.5
Differential diagnosis. Administering naloxone comes with many risks. If a patient develops shortness of breath after receiving naloxone, one should have clinical suspicion for the formation of pulmonary edema. However, ruling out other potential differential diagnoses is important. Pulmonary edema can result from an allergic reaction, cardiac issue, fluid overload, or reaction to naloxone.15,19-21 In our patient, the lack of rash or hives ruled out an allergic reaction. Pre- and post-operative cardiac examinations did not reveal a murmur, and results of a telemetry electrocardiogram (EKG) scan did not raise concern for arrhythmia; thus, cardiac issues were unlikely.
Our patient received 293 mL of lactated Ringer's solution intraoperatively. Given his weight of 60 kg, this amount of fluid was only 4.9 mL/kg. A standard normal saline bolus would be 20 mL/kg. Thus, his intraoperative fluid administration would not account for his symptoms, and fluid overload was unlikely. Additionally, the patient was not given any oral medications postoperatively to suggest aspiration. Given the temporal correlation of the administration of naloxone and the development of pink frothy sputum, the patient's clinical status was likely due to naloxone administration.
A wide range of doses of naloxone (0.04 mg-0.4 mg) are implicated in naloxone-induced negative pressure pulmonary edema.4,22 Our patient received multiple 40 mcg doses, totaling 200 mcg (0.2 mg); whereas he could have received a max dose ranging from 0.3 mg to 0.9 mg of naloxone to help reverse his respiratory depression secondary to therapeutic opioid dosing. This demonstrates that small doses of naloxone can be implicated in the development of pulmonary edema.
A review of the patient’s vital signs demonstrated that his blood pressure was higher prior to naloxone administration compared with after administration. Additionally, there was only an increase of 5 bpm in heart rate after receiving naloxone. However, hemodynamic changes secondary to the catecholamine surge that results in neurogenic pulmonary edema can develop within seconds. These changes were likely not detected during the automated vital sign assessments. The patient started to cough and had pink frothy sputum only after receiving naloxone. There was no mention of the patient having stridor or signs of increased breathing immediately after extubation or after receiving naloxone to suggest that the patient experienced laryngospasm.
This data demonstrates that development of pulmonary edema in this case was unlikely due to a negative pressure development secondary to laryngospasms but may have been secondary to a catecholamine surge that primarily affected the pulmonary vasculature. This mechanism has been proposed by several previous studies.18,23
Treatment and management. The treatment for pulmonary edema (whether induced by naloxone or not) is generally supportive. Use of noninvasive positive pressure ventilation—either by continuous positive airway pressure (CPAP) or BiPAP—can be helpful in mitigating continued effects of pulmonary edema. However, there may be times where reintubation is necessary.7,11 Mixed reviews as to whether diuretics may be helpful in the clinical management of pulmonary edema secondary to negative pressure development are found in the literature.24,25 Our patient was given a single dose of furosemide prior to reintubation.
Conclusions. Pulmonary edema formation should be considered in patients experiencing respiratory distress after being extubated and in emergence from general anesthesia. Although multiple mechanisms for pulmonary edema development are possible, naloxone should be considered as a cause for the pulmonary edema if there is a temporal association between infusion of the naloxone and the development of pink frothy sputum after extubation. Generally, supportive care is needed; however, positive pressure ventilation or reintubation may be required for respiratory care.
Patient outcome. In our case, the patient was noted to have blood-tinged sputum after he was placed on BiPAP. A decision was made to reintubate him. Subsequently, his PEEP increased from 8 to 10 cmH2O to help mitigate further losses and help improve aeration. He remained intubated for a total of 2 days, after which he was slowly weaned off the ventilator to a nasal cannula, and then to room air. After receiving mechanical ventilation with enhanced PEEP, his chest radiography scans demonstrated overall improvement (Figure 2).
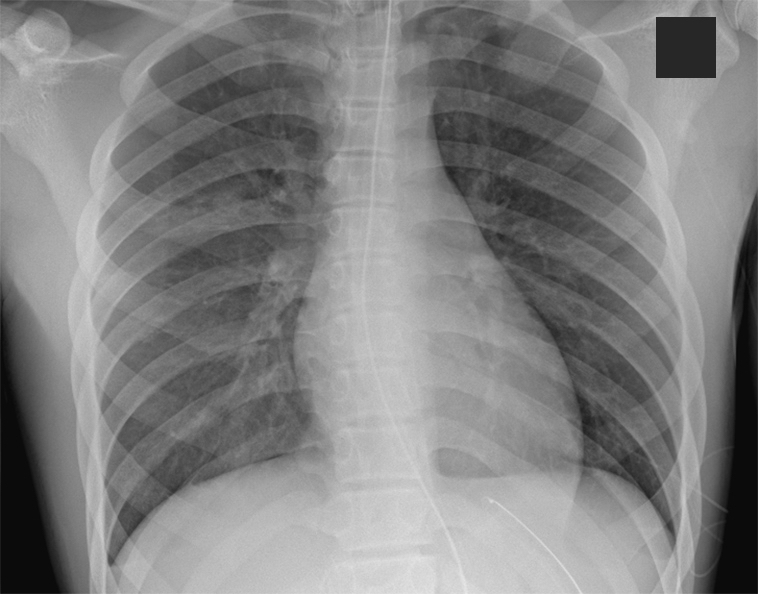
Figure 2. Improvement of pulmonary edema after mechanical ventilation.
References
- Wermeling DP. Review of naloxone safety for opioid overdose: practical considerations for new technology and expanded public access. Ther Adv Drug Saf. 2015;6(1):20-31. https://doi.org/10.1177/2042098614564776
- Schwartz JA, Koenigsberg MD. Naloxone-induced pulmonary edema. Ann Emerg Med. 1987;16(11):1294-1296. https://doi.org/10.1016/s0196-0644(87)80244-5
- Horng HC, Ho MT, Huang CH, Yeh CC, Cherng CH. Negative pressure pulmonary edema following naloxone administration in a patient with fentanyl-induced respiratory depression. Acta Anaesthesiol Taiwan. 2010;48(3):155-157. https://doi.org/10.1016/s1875-4597(10)60050-1
- Flacke JW, Flacke WE, Williams GD. Acute pulmonary edema following naloxone reversal of high-dose morphine anesthesia. Anesthesiology. 1977;47(4):376-378. https://doi.org/10.1097/00000542-197710000-00009
- Bansal S, Khan R, Tietjen PA. Naloxone-induced pulmonary edema. Chest. 2007;132(4_Meeting Abstracts):692. doi:10.1378/chest.132.4_MeetingAbstracts.692
- Andree RA. Sudden death following naloxone administration. Anesth Analg. 1980;59(10):782-784. https://journals.lww.com/anesthesia-analgesia/Citation/1980/10000/Sudden_Death_following_Naloxone_Administration.12.aspx
- Thiagarajan RR, Laussen PC. Negative pressure pulmonary edema in children—pathogenesis and clinical management. Paediatr Anaesth. 2007;17(4):307-310. https://doi.org/10.1111/j.1460-9592.2007.02197.x
- Cascade PN, Alexander GD, Mackie DS. Negative-pressure pulmonary edema after endotracheal intubation. Radiology. 1993;186(3):671-675. https://doi.org/10.1148/radiology.186.3.8430172
- Toukan Y, Gur M, Bentur L. Negative pressure pulmonary edema following choking on a cookie. Pediatr Pulmonol. 2016;51(7):E25-E27. https://doi.org/10.1002/ppul.23385
- Domnica PJ, Floret D, Bouchut JC. Negative pressure pulmonary edema and airway foreign body retrieval: anesthetic considerations. Paediatr Anaesth. 2010;20(3):288-289. https://doi.org/10.1111/j.1460-9592.2009.03229.x
- McConkey PP. Postobstructive pulmonary oedema—a case series and review. Anaesth Intensive Care. 2000;28(1):72-76. https://doi.org/10.1177/0310057x0002800114
- Johnson C, Mayer P, Grosz D. Pulmonary edema following naloxone administration in a healthy orthopedic patient. J Clin Anesth. 1995;7(4):356-357. https://doi.org/10.1016/0952-8180(95)00038-j
- Tanaka GY. Letter: hypertensive reaction to naloxone. JAMA. 1974;228(1):25-26. https://doi.org/10.1001/jama.1974.03230260019008
- Freye E. Cardiovascular effects of high dosages of fentanyl, meperdine, and naloxone in dogs. Anesth Analg. 1974;53(1):40-47. https://journals.lww.com/anesthesia-analgesia/Abstract/1974/01000/Cardiovascular_Effects_of_High_Dosages_of.10.aspx
- Taff RH. Pulmonary edema following naloxone administration in a patient without heart disease. Anesthesiology. 1983;59(6):576-577. https://doi.org/10.1097/00000542-198312000-00019
- Dahan A, Aarts L, Smith TW. Incidence, reversal, and prevention of opioid-induced respiratory depression. Anesthesiology. 2010;112(1):226-238. https://doi.org/10.1097/aln.0b013e3181c38c25
- Theodore J, Robin ED. Pathogenesis of neurogenic pulmonary oedema. Lancet. 1975;2(7938):749-751. https://doi.org/10.1016/s0140-6736(75)90729-1
- Prough DS, Roy R, Bumgarner J, Shannon G. Acute pulmonary edema in healthy teenagers following conservative doses of intravenous naloxone. Anesthesiology. 1984;60(5):485-486. https://doi.org/10.1097/00000542-198405000-00018
- Solomon DR. Anaphylactoid reaction and non-cardiac pulmonary edema following intravenous contrast injection. Am J Emerg Med. 1986;4(2):146-149. https://doi.org/10.1016/0735-6757(86)90161-0
- Ingbar DH. Cardiogenic pulmonary edema: mechanisms and treatment - an intensivist's view. Curr Opin Crit Care. 2019;25(4):371-378. https://doi.org/10.1097/mcc.0000000000000626
- Claure-Del Granado R, Mehta RL. Fluid overload in the ICU: evaluation and management. BMC Nephrol. 2016;17(1):109. https://doi.org/10.1186/s12882-016-0323-6
- Partridge BL, Ward CF. Pulmonary edema following low-dose naloxone administration. Anesthesiology. 1986;65(6):709-710. https://doi.org/10.1097/00000542-198612000-00037
- Joyce JA. AANA journal course: update for nurse anesthetists—anesthesia-related noncardiogenic pulmonary edema: a literature review. AANA J. 1999;67(5):469-473.
- Maxwell BG, Mihm FG. Questioning diuretic use in acute negative-pressure pulmonary edema. Anesthesiology. 2011;114(2):461. https://doi.org/10.1097/aln.0b013e31820708c4
- Krodel DJ, Bittner EA, Abdulnour R, Brown R, Eikermann M. Case scenario: acute postoperative negative pressure pulmonary edema. Anesthesiology. 2010;113(1):200-207. https://doi.org/10.1097/aln.0b013e3181e32e68


