Peer Reviewed
Osteomalacia: Case Report and Literature Review
Authors:
Asha Patnaik, MD
Assistant Professor of Clinical Medicine, Division of Rheumatology, Renaissance School of Medicine at Stony Brook University, Stony Brook, New York
Barjinder Buttar, MD
Internal Medicine Residency Program, Zucker School of Medicine/Northwell Health at Mather Hospital, Port Jefferson, New York
Michael Malekan, DO
Fellow in Rheumatology, Renaissance School of Medicine at Stony Brook University, Stony Brook, New York
Saleha Riaz, DO
Fellow in Rheumatology, Renaissance School of Medicine at Stony Brook University, Stony Brook, New York
Igor Kravets, MD
Associate Professor of Clinical Medicine, Division of Endocrinology, Renaissance School of Medicine at Stony Brook University, Stony Brook, New York
Citation:
Patnaik A, Buttar B, Malekan M, Riaz S, Kravets I. Osteomalacia: case report and literature review. Consultant. 2019;59(9):272-274.
A 38-year-old man was admitted to the hospital with a comminuted right intertrochanteric hip fracture (Figure 1) after having experienced a minor fall while walking down a few steps at home. He had a history of uncontrolled type 2 diabetes mellitus and early-onset chronic kidney disease (CKD) secondary to diabetic nephropathy. On admission, radiography findings were significant for multiple healing right anterior rib fractures. The patient underwent an open reduction and internal fixation of the right hip with placement of an intramedullary nail (Figure 2).
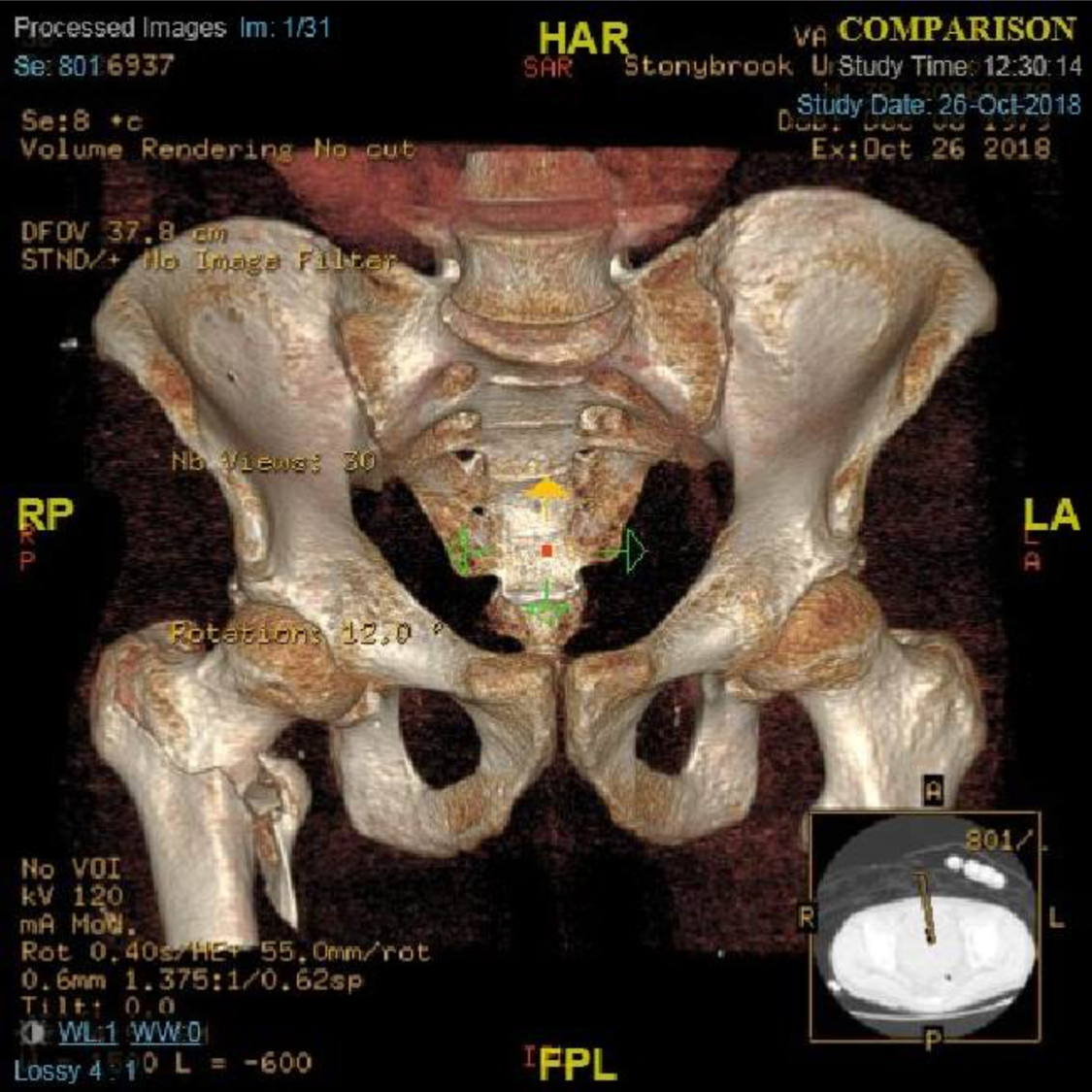
Figure 1. A computed tomography scan of the pelvis with bony reformats showing an acute, comminuted, mildly displaced and mildly impacted intertrochanteric fracture of the proximal right femur.
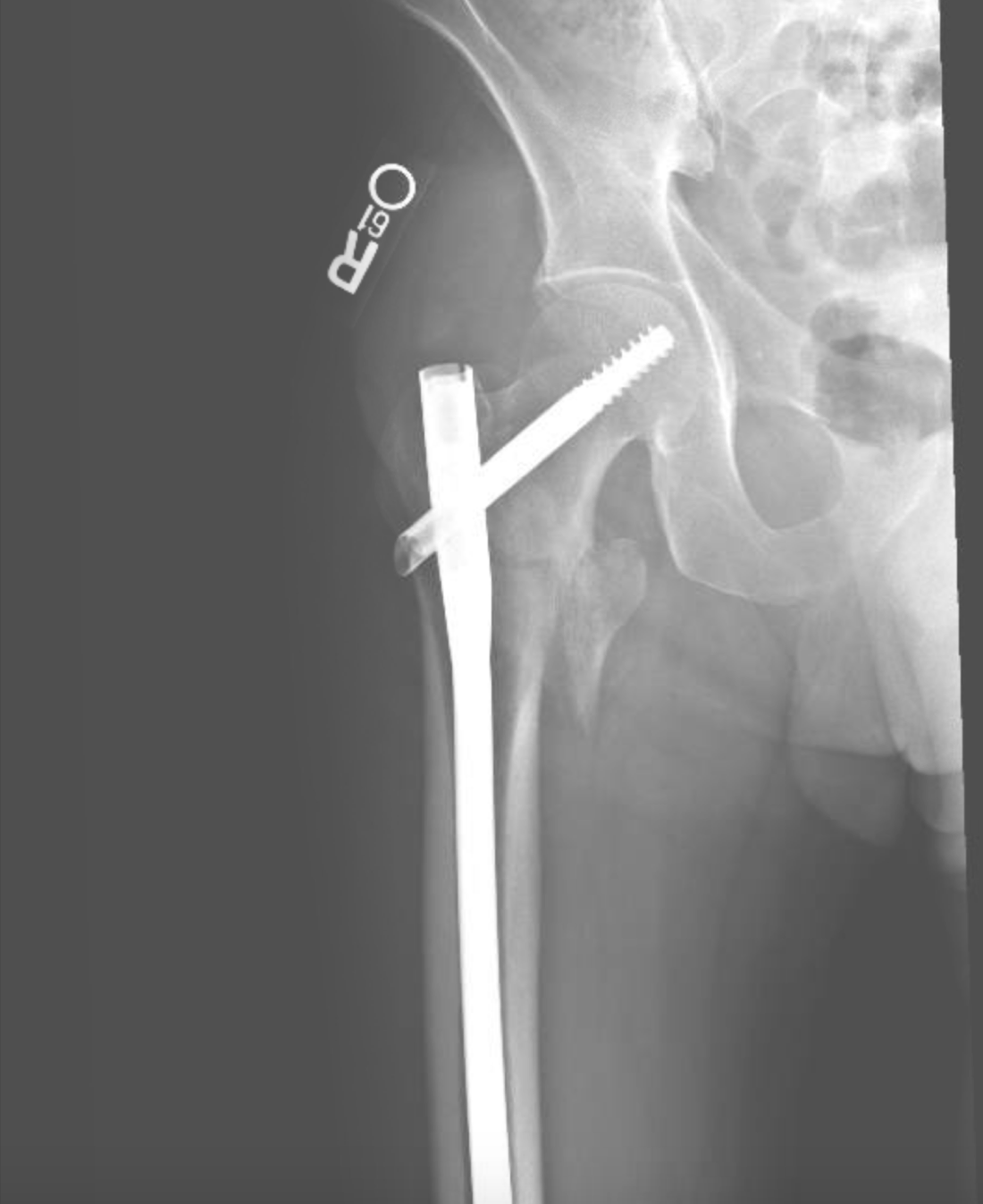
Figure 2. A postsurgical radiograph showing placement of a cephalocaudal medullary long nail with sliding hip screw.
An endocrinologist was consulted to evaluate the patient for a possible mineral and bone disorder. The patient was found to have a severe vitamin D deficiency with a 25-hydroxyvitamin D level of 6 ng/mL (reference range, 20–100 ng/mL). Serum levels of calcium, phosphorus, alkaline phosphatase, and intact parathyroid hormone (PTH), as well as thyroid function, were all within normal limits.
A diagnosis of adult-onset osteomalacia was made based upon his clinical presentation, radiographic findings, and isolated vitamin D deficiency. The patient was started on oral ergocalciferol, 50,000 IU, weekly for 4 weeks and was instructed to follow up for continued treatment and monitoring.
Discussion. CKD has been recognized as a growing international health problem. The Kidney Disease: Improving Global Outcomes (KDIGO) group was established in 2003 to provide continued improvement in the care and diagnosis of persons with CKD worldwide. In 2006, the group defined the clinical syndrome chronic kidney disease–mineral and bone disorder (CKD-MBD) in an effort to improve communication, facilitate clinical decision-making, and allow for the development of future evidence-based clinical practice guidelines.
The syndrome CKD-MBD is defined as the systemic manifestations of all mineral and bone metabolism disorders that occur in the setting of CKD. These disorders include osteomalacia, osteitis fibrosa cystica, adynamic bone disease, and mixed uremic type (Table).1,2
|
Table. Classification of CKD-MBD1,2 |
|||
|
Osteomalacia |
Osteitis Fibrosa Cystica |
Adynamic Bone Disease |
Mixed Uremic Type |
|
Low bone turnover |
High bone turnover |
Low bone turnover |
High or low bone turnover |
|
Abnormal mineralization |
Normal mineralization |
Normal mineralization |
Abnormal mineralization |
|
Variable bone volume depending on duration of disease |
Variable bone volume depending on duration of disease |
Low bone volume |
Normal bone volume |
|
Softening of bone due to deficiency in calcitriol and free ionized calcium |
Weakening of bones due to high turnover and replacement of bony support structures with fibrous tissue |
Reduced osteoblast and osteoclast activity with markedly low bone turnover |
Either high or low bone turnover and persistent mineralization defect |
|
Caused by aluminum deposited in bone secondary to use of aluminum-based phosphate binders |
Caused by persistent excess of PTH leading to activation of osteoclasts and breakdown of bone |
Caused by suppression of PTH in treated CKD patients, especially those on dialysis |
Caused by persistent vitamin D deficiency |
Osteomalacia develops as a direct result of the inability to absorb and process calcium, phosphorus, and vitamin D. Effective bone mineralization depends on vitamin D metabolism, which starts with sunlight converting 7-dehydrocholesterol to cholecalciferol (vitamin D3). Cholecalciferol is then hydroxylated in the liver by the enzyme 25-hydroxylase to 25-hydroxyvitamin D. The enzyme 1α-hydroxylase will further hydroxylate 25-hydroxyvitamin D to 1,25-dihydroxyvitamin D (calcitriol). Calcitriol, the active form of vitamin D, finally binds to receptors in the intestines to promote absorption of calcium and phosphorus.3 Defects in this pathway lead to reduced absorption and metabolism of calcium and vitamin D.
Decreased serum calcium levels result in an increase in the secretion of PTH. A rise in PTH will directly increase the reabsorption of calcium from bone by stimulating osteoclast activity. The goal of this feedback mechanism is to bring the serum calcium level back up to a normal range. In osteomalacia, however, the serum calcium level remains persistently low, resulting in a continuous secretion of PTH and the development of secondary hyperparathyroidism. Elevated PTH leads to hypophosphatemia by reducing the reabsorption of phosphate at the proximal tubule of the kidneys.4
Osteomalacia causes progressive softening of bone due to decreased mineralization of newly formed osteoid at bone turnover sites. Osteoid refers to bone protein matrix, which is mainly composed of type 1 collagen.5 The most common causes of osteomalacia are vitamin D deficiency secondary to an inadequate diet, lack of sun exposure, and gastrointestinal tract conditions leading to fat malabsorption.6 In patients with CKD, the kidneys have a reduced ability to synthesize calcitriol; the same is true for chronic liver failure, in which ongoing damage reduces the liver’s ability to hydroxylate vitamin D.7
Many medications can lead to the breakdown of vitamin D and its metabolites. Antibiotics such as rifampicin, excessive intake of aluminum-based antacids, and anticonvulsants including carbamazepine, phenytoin, and sodium valproate are a few examples. Aluminum-based phosphate binders, used to treat hyperphosphatemia in patients with advanced CKD receiving hemodialysis, cause osteomalacia through toxic aluminum deposition in bone.8
Clinical manifestations of osteomalacia include bone pain, fractures, muscle weakness, spasms, and cramps. Fractures most commonly occur in the lower extremities, the lower spine, and the pelvis.9
Laboratory abnormalities include elevated alkaline phosphatase, elevated intact PTH, reduced serum calcium and phosphorus, and reduced 25-hydroxyvitamin D.10
Radiographic imaging of patients with osteomalacia will show reduced bone mineral density with thinning of the cortex. As the disease progresses, softening of the bone leads to the development of biconcave vertebral bodies referred to as “codfish” or “fish-mouth” vertebrae.10 Also seen are Looser zones, referred to as cortical infractions or Milkman lines. These zones are most commonly identified on radiographs at the public rami, medial proximal femur, lateral scapula, posterior proximal ulna, and the ribs. They are seen as narrow radiolucent lines with sclerotic borders and mimic fractures and fissures.10
The gold standard for diagnosing osteomalacia is a bone biopsy with tetracycline double-labeling.11 Tetracycline is given both 3 weeks before and 3 to 5 days before the bone biopsy. Tetracycline binds to hydroxyapatite and labels bone, which can then be visualized by fluorescence microscopy. The amount of bone formed between the tetracycline signals is used to calculate bone turnover.11 Bone biopsy remains the gold standard for diagnosis, because traditional radiologic and serum testing cannot reliably differentiate between different bone turnover diseases. To interpret results of a bone biopsy, the TMV system is used.1 This system evaluates three key histologic descriptors: bone turnover (T), mineralization (M), and volume (V). Bone turnover and volume is described as high, normal, or low while bone mineralization is normal or abnormal.1
Due to the cost and invasiveness of bone biopsies, the practical time delays, and the interpretation issues, they are rarely performed for diagnostic confirmation. As a result, new advancements in diagnosing osteomalacia focus on noninvasive approaches.2 Dual energy x-ray absorptiometry, trabecular bone score (TBS), conventional quantitative computed tomography (QCT), high-resolution peripheral QCT, and micro magnetic resonance imaging are a few examples.2 These imaging options can assess bone density and structural aspects of bone quality.2 In addition, bone turnover markers assess aspects of bone quality that cannot be evaluated by way of imaging, such as bone formation rates and mineralization.2
The goal of osteomalacia treatment is to address the underlying disorder and to correct vitamin D deficiency, hypocalcemia, and hypophosphatemia. Adequate supplementation will lead to significant improvements in muscle strength and bone tenderness within a few weeks to a month. Bone mineral density also will improve over 3 to 6 months.12 It is important to regularly monitor serum calcium and urinary calcium excretion of patients after the first and third month, and then every 6 to 12 months thereafter. Monitoring can be discontinued safely once the 24-hour urinary calcium excretion is within normal limits. Serum 25-hydroxyvitamin D should be measured every 3 to 4 months, as well, and the supplementation dose should be adjusted accordingly.12 An appropriate treatment regimen for severe vitamin D deficiency with a serum 25-hydroxyvitamin D concentration less than 20 ng/mL is 50,000 IU of vitamin D2 or D3 orally once per week for 6 to 8 weeks, followed by 800 IU of vitamin D2 daily thereafter.13 All patients should also consume a total calcium of at least 1000 to 1200 mg per day.13
References:
- Moe S, Drüeke T, Cunningham J, et al. Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2006;69(11):1945-1953.
- Damasiewicz MJ, Nickolas TL. Rethinking bone disease in kidney disease. JBMR Plus. 2018;2(6):309-32
- Walker J. Pathogenesis, diagnosis, and management of osteomalacia. Nurs Older People. 2014;26(6):32-37.
- Ralston SH, McInnes LB. Rheumatology and bone disease. In: Walker BR, Colledge NR, Ralston SH, Penman ID, eds. Davidson’s Principles and Practice of Medicine. 22nd ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2014:chap 25.
- Sitta MdC, Cassis SVA, Horie NC, Moyses RMA, Jorgetti V, Garcez-Leme LE. Osteomalacia and vitamin D deficiency in the elderly. Clinics (Sao Paulo). 2009;64(2):156-158.
- Noh C-K, Lee M-J, Kim BK, Chung Y-S. A case of nutritional osteomalacia in a young adult male. J Bone Metab. 2013;20(1):51-55.
- Williams S, Malatesta K, Norris K. Vitamin D and chronic kidney disease. Ethn Dis. 2019;19(4 suppl 5):S5-8–S5-11.
- Robien K, Oppeneer SJ, Kelly JA, Hamilton-Reeves JM. Drug–vitamin D interactions: a systematic review of the literature. Nutr Clin Pract. 2013;28(2):194-20
- Hazzaizi MA, Alzeer I, Tamimi W, Al Atawi M, Al Alwan I. Clinical presentation and etiology of osteomalacia/rickets in adolescents. Saudi J Kidney Dis Transpl. 2013;24(5):938-941.
- Sahay M, Sahay R. Rickets–vitamin D deficiency and dependency. Indian J Endocrinol Metab. 2012;16(2):164-176.
- Moorthi RN, Moe SM. CKD–mineral and bone disorder: core curriculum 20 Am J Kidney Dis. 2011;58(6):1022-1036.
- Cohen A, Drake MT. Clinical manifestations, diagnosis, and treatment of osteomalacia. UpToDate. https://www.uptodate.com/contents/clinical-manifestations-diagnosis-and-treatment-of-osteomalacia. Updated July 1, 2019. Accessed July 16, 2019.
- Holick MF. The vitamin D deficiency pandemic: approaches for diagnosis, treatment and prevention. Rev Endocr Metab Dis. 2017;18(2):153-165.


