Peer Reviewed
Oral Albuterol Overdose With Electrocardiographic Changes and Cardiac Ischemia
Authors:
Teresa D. Frey, DO; William F. Waltz, MD, PhD; and Jody N. Huber, MD
Sanford USD Medical Center, Sanford Children’s Hospital, Department of Pediatrics, Sioux Falls, South Dakota; and University of South Dakota Sanford School of Medicine, Vermillion, South Dakota
Citation:
Frey TD, Waltz WF, Huber JN. Oral albuterol overdose with electrocardiographic changes and cardiac ischemia. Consultant. 2019;59(11):343-344.
A 15-year-old girl with a history of depression was brought to the emergency department (ED) by emergency medical services (EMS) for nausea and vomiting. Five hours prior to presentation, she had intentionally taken 30 8-mg albuterol sulfate tablets that had been prescribed to her mother more than 10 years previously. The adolescent quickly became symptomatic and reported the suicide attempt to a friend, who called EMS.
At presentation to the ED, the patient’s vital signs were as follows: heart rate, 167 beats/min; blood pressure, 99/72 mm Hg; temperature, 37.2°C; and respiratory rate, 24 breaths/min. She reported having chest pain and palpitations.
Results of initial laboratory tests revealed hyperglycemia (glucose, 282 mg/dL), hypokalemia (potassium, 2.5 mEq/L), leukocytosis (white blood cell count, 26,100/µL), and an elevated troponin level (0.07 ng/mL). She was transferred to a tertiary care facility.
Upon admission to the pediatric intensive care unit, her vital signs were as follows: heart rate, 161 beats/min; blood pressure, 83/37 mm Hg; temperature, 37.2°C; and respiratory rate, 27 breaths/min. Physical examination findings were remarkable for diaphoresis, mydriasis, dry mucous membranes, and a hyperdynamic precordium with a grade 2/6 systolic ejection murmur.
Repeated laboratory tests demonstrated a troponin level further elevated to 0.25 ng/mL, a potassium level of 2.3 mEq/L, a calcium level of 7.9 mg/dL, and glucose level of 304 mg/dL. She received 3 L of normal saline and 1 g of calcium gluconate without improvement in hypotension. Esmolol was initiated to slow the heart rate, with titration to 300 µg/kg/min. Her tachycardia subsequently improved to 150 beats/min; however, the hypotension persisted, requiring initiation of a phenylephrine infusion at 100 µg/min. In addition, she received a total of 40 mEq of potassium chloride by intravenous infusion.
Initial electrocardiography (ECG) findings were concerning for ST-segment depression with possible prolonged QTc, sinus tachycardia, interventricular conduction delay, and bilateral atrial enlargement (Figure 1)
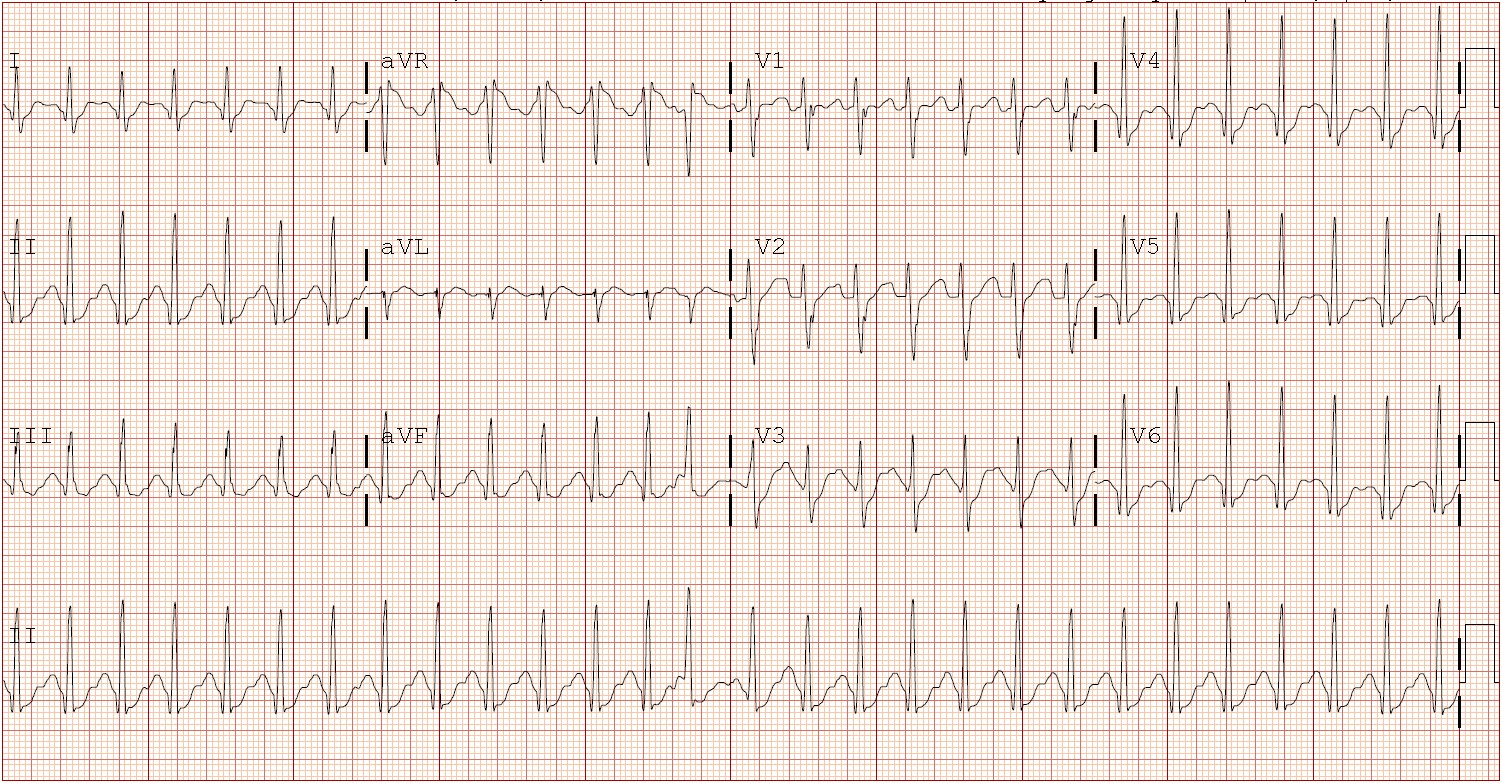
Echocardiography revealed normal cardiac anatomy that was hyperdynamic, with an ejection fraction of 68.3% and bilateral diastolic dysfunction of the ventricles. The diastolic hypotension nadir was 26 mm Hg, and the troponin level peaked at 0.60 ng/mL on hospital day 1. Nitroglycerin and a nasal cannula were initiated to improve coronary perfusion and prevent further cardiac ischemia. Repeated ECGs showed improvement, with resolution of the ST-depression (Figure 2).

She was weaned from phenylephrine, esmolol, and nitroglycerin by hospital day 2. Laboratory test abnormalities, including hypokalemia and hyperglycemia, resolved. She was discharged on hospital day 5 to an inpatient mental health facility for further management of depression.
The patient was reevaluated at a cardiology clinic 15 days after admission. At that time, she was asymptomatic, with normal echocardiography findings, a troponin level less than 0.01 ng/mL, and a brain-type natriuretic peptide level of 11 pg/mL. An exercise stress test with myocardial perfusion was performed approximately 2 months after admission, the results of which were positive for a small amount of reversible ischemia in the anterior wall at the level of the apex. She will undergo a repeated exercise stress test and myocardial perfusion study 1 year after the hospital admission. It was decided to allow her to participate in physical activity with no further precautions.
Discussion. Historically, albuterol use in pediatric patients is safe, with most of its effects occurring at β2 receptors in the lungs, since the most common method of delivery is inhaled. This patient’s case demonstrates that, if ingested systemically, albuterol can have significant systemic effects, as well.
Albuterol is a sympathomimetic agent that can cause cardiac abnormalities by relaxing vascular smooth muscle via β2 receptors in blood vessels, causing diastolic hypotension.1 While diastolic hypotension is rare with inhaled intermittent albuterol, there are reports that it is correlated with the dose and age of the child. Therefore, individuals who are older and receive higher doses of albuterol are more likely to have hypotension.1 Individuals who receive continuous inhaled albuterol will often have hypotension and ST-segment abnormalities.
Oral liquid albuterol had been widely used, but associated adverse effects were more prevalent. Oral systemic versions are now rarely used and have been replaced by the safer, inhaled intermittent albuterol. More commonly seen adverse cardiac effects include tachycardia and increased contractility. While these effects are not particularly harmful to most patients, they can worsen cardiac dysfunction. Tachycardia will lead to decreased perfusion of the myocardium and decreased time in diastole, causing cardiac ischemia and potentially irreversible myocardial damage.1 Hypokalemia is a well-known adverse effect of β2 agonists and is thought to be secondary to the stimulation of sodium-potassium-ATPase channels and subsequent shifting of potassium intracellularly. This is dose-dependent and should be monitored closely, especially in patients on continuous or high-dose β2 agonists.2
While many case reports have documented hypokalemia associated with albuterol toxicity, conduction abnormalities and cardiac ischemia are extremely rare. Hyperglycemia can be seen initially, which is secondary to stimulation of β2 receptors in the pancreas and increased glycogenolysis in muscles and/or the liver. This hyperglycemia often is transient but leads the body to respond by producing large amounts of insulin and, subsequently, a delayed hypoglycemia.3 These adverse effects can be seen as quickly as 30 minutes, with a reported half-life of 8 hours, and often continue up to 16 hours after the ingestion.4
Take-home message. Albuterol is a widely used medication in pediatric and adult populations. However, oral albuterol is rarely used today due to improved methods of delivery and concern for cardiac toxicity. Not only is this an unusual medication in today’s pharmacopeia, but little evidence exists on the optimal treatment of patients with oral β2 agonist toxicity. While many of the adverse effects are not life-threatening, this case report demonstrates that high doses or systemic use can lead to severe, long-term sequelae.
References:
- Sarnaik SM, Saladino RA, Manole M, et al. Diastolic hypotension is an unrecognized risk factor for β-agonist-associated myocardial injury in children with asthma. Pediatr Crit Care Med. 2013;14(6):e273-e279.
- Lewis LD, Essex E, Volans GN, Cochrane GM. A study of self poisoning with oral salbutamol—laboratory and clinical features. Hum Exp Toxicol. 1993;12(5):397-401.
- Ozdemir D, Yilmaz E, Duman M, Unal N, Tuncok Y. Hypoglycemia after albuterol overdose in a pediatric patient. Pediatr Emerg Care. 2004;20(7):464-465.
- Glatstein MM, Rimon A, Koren L, Marom R, Danino D, Scolnik D. Unintentional oral beta agonist overdose: case report and review of the literature. Am J Ther. 2013;20(3):311-31


