Peer Reviewed
Klippel-Trénaunay Syndrome
Authors:
Shariar Akter, MD
McGovern Medical School at UT Health, Houston, Texas
Nina Lahoti
Rice University, Houston Texas
Lynnette Mazur, MD, MPH
McGovern Medical School at UT Health, Houston, Texas
Citation:
Akter S, Lahoti N, Mazur L. Klippel-Trénaunay syndrome. Consultant. 2019;59(7):220-222.
A newborn boy was noted to have a diffuse erythematous rash at birth. He had been born at 40 weeks of gestation to a 19-year-old gravida 2, para 2 mother who had had adequate prenatal care. The pregnancy had been complicated by chlamydia infection with documented cure after treatment, anemia, and an episode of self-limited hives. He had been born by spontaneous vaginal delivery and had Apgar scores of 8 at 1 minute and 9 at 5 minutes.
At birth, his weight, length, and head circumference were 3.87 kg (85th percentile), 51 cm (72nd percentile), and 35 cm (66th percentile), respectively. Physical examination findings were remarkable for a diffuse erythematous macular rash with blanching on his trunk and extremities (Figures 1 and 2). There was no facial involvement, and the palms and soles also were spared. No nevi or mongolian spots were noted.
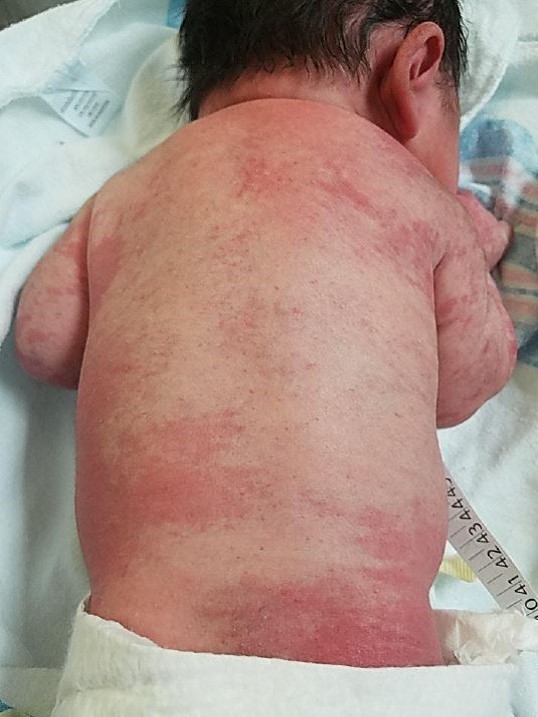
Figure 1. A diffuse erythematous rash on the newborn’s trunk.
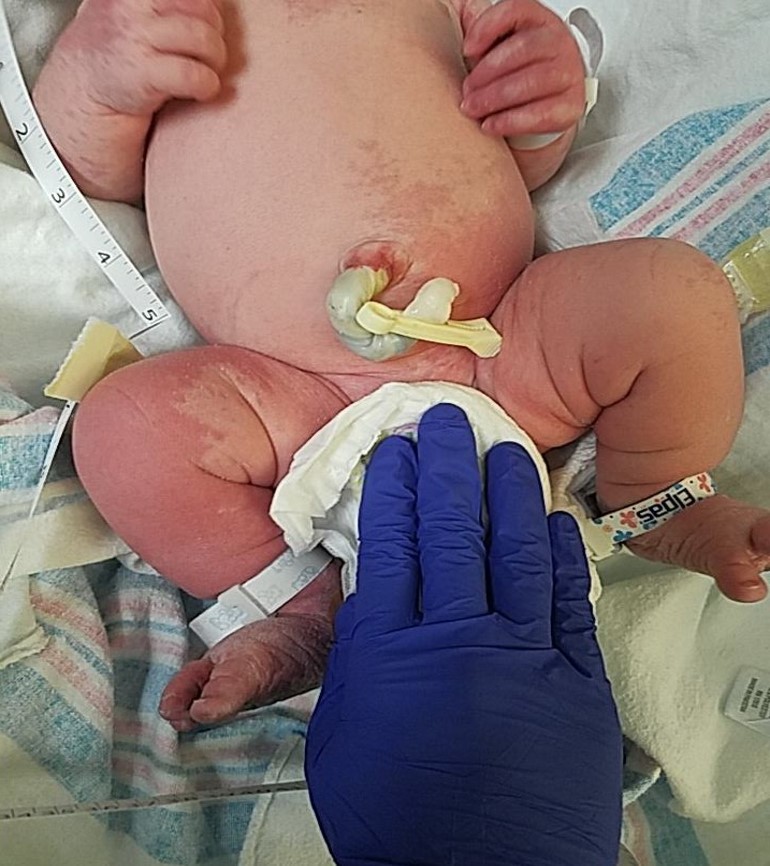
Figure 2. Erythematous, poorly demarcated macules, primarily on the trunk and lower extremities with some upper-extremity involvement but sparing the face.
The differential diagnosis included both infectious and vascular anomalies (Table). Results of a complete blood count were unremarkable, with the exception of a low platelet count of 60 × 103/µL.
|
Table. Differential Diagnosis of Vascular Rash in Newborn |
||
|
Diagnosis |
Skin Findings |
Other/Complications |
|
Capillary malformation (port-wine stain)/arteriovenous malformation1 |
Erythematous macular stains, pink to dark red |
Associated with Sturge-Weber syndrome (face), Klippel-Trénaunay syndrome, Parkes Weber syndrome, phakomatosis pigmentovascularis, Proteus syndrome, Cobb syndrome, Bannayan-Riley-Ruvalcaba syndrome, Beckwith-Wiedemann syndrome, von Hippel-Lindau disease, Rubinstein-Taybi syndrome, Wyburn-Mason syndrome, Roberts syndrome, Coat disease, pyogenic granuloma |
|
Congenital cutaneous candidiasis2 |
Erythematous macules, papules, pustules, and bullae; diffuse distribution, (palms and soles); well-defined borders |
Nail changes (yellow discoloration, thickening, paronychia) |
|
Cutis marmorata telangiectatica congenita3 |
Erythematous or violaceous reticulated or marbled patches (localized or generalized); no improvement with warming |
Macrocephaly, hydrocephalus, seizures, developmental delay, glaucoma, retinal detachment, limb hypoplasia/hyperplasia on the affected side |
|
Kaposiform hemangioendothelioma1 |
Benign subcutaneous blood tumors; violaceous subcutaneous nodules |
Thrombocytopenia, anemia, elevated D-dimer level, fragmented erythrocytes on smear |
|
Klippel-Trénaunay syndrome1 |
Erythematous, violaceous macules; triad of vascular malformation, venous varicosity, and hyperplasia of soft tissue and bone |
Macro/microcephaly, facial hypertrophy, intracranial calcifications, seizures, developmental delay, glaucoma, cataracts, pulmonary emboli, congestive heart failure, limb hypertrophy, scoliosis, hip dislocation, lymphedema, ulcerations |
|
Parkes Weber syndrome1 |
Arteriovenous malformations, in addition to the findings in Klippel-Trénaunay syndrome |
Abnormal ear shape, coarctation of the aorta, in addition to the findings Klippel-Trénaunay syndrome |
|
Psoriasis3 |
Erythematous, well-marginated plaques with silvery scales; bilateral and symmetric |
Obesity, cardiovascular problems (hypertension), diabetes mellitus |
The newborn was admitted to the high-risk nursery for evaluation of sepsis due to his rash and thrombocytopenia. Ampicillin and gentamycin were continued until blood cultures were negative at 60 hours of life. A dermatologist was consulted, and skin cultures were obtained for suspected candida or other fungal infections, the results of which were eventually negative. On day of life 3, he was discharged home with a diagnosis of cutis marmorata telangiectatica congenita and was scheduled for a dermatology follow-up.
At his 2-month well-child visit, the rash remained unchanged. State newborn screening test results were normal.
At 3 months of age, the mother returned to the clinic, concerned that the infant’s leg was larger than his right (2.5 cm difference in girth). An ultrasonogram obtained in the emergency department revealed dampened arterial flow within the left anterior and posterior tibial arteries. The vascular surgery team recommended a magnetic resonance angiogram (MRA), which revealed extensive collateral vessels, predominantly in the thighs and knees, and turbulent flow within the common femoral and superficial femoral veins (Figure 3). All vessels were otherwise patent, and no thromboemboli were noted. Abdominal ultrasonography findings excluded other vascular abnormalities.
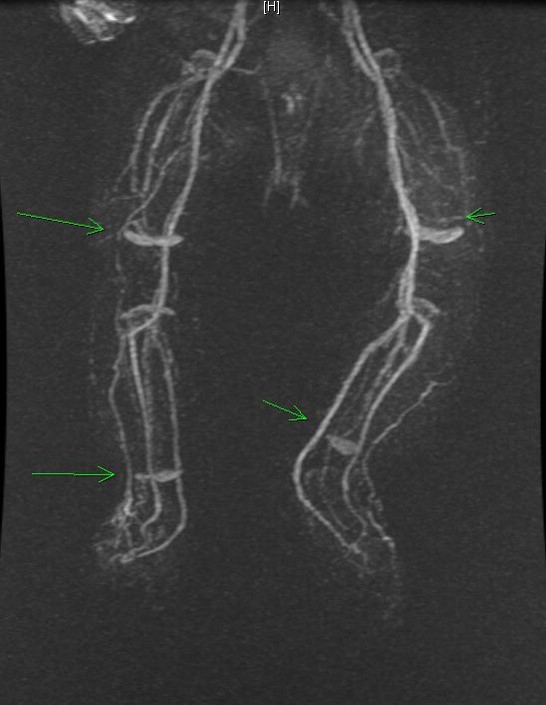
Figure 3. An MRA of the lower extremities showed evidence of significant collateral blood vessel formation, primarily at the thighs and knees, with some in the lower leg.
Based on the rash, the venous malformations identified on imaging, and the limb hypertrophy, he received a diagnosis of Klippel-Trénaunay syndrome (KTS) and was referred to the vascular clinic.
DISCUSSION
Port-wine stains (PWS), also known as capillary malformations, may occur as isolated lesions or in association with a variety of malformations. One such association is with KTS, also called angio-osteohypertrophy syndrome or hemangiectatic hypertrophy. KTS is a congenital abnormality characterized by PWS and venous and lymphatic malformations with localized (soft tissue and bony hypertrophy) overgrowth of the affected extremity.4,5 There is evidence that KTS may be associated with a somatic mutation of PIK3CA (the phosphatidylinositol-4,5-bisphosphonate 3-kinase catalytic subunit alpha gene).6 Although our patient had PWS, his skin lesions were not sharply demarcated, affected an unusually large area (between 25% and 30%, of his body), and involved more than 1 extremity.
Complications of KTS can affect multiple body systems and warrant a multidisciplinary approach to management. Our patient is at increased risk for venous thromboemboli, which can occur in up to 40% of patients with KTS.7,8 Although the exact mechanism is unknown, these may result from venous stagnation and altered flow within the distorted and enlarged blood vessels, which leads to hypercoagulability. One study showed that patients with KTS have higher plasma levels of D-dimer and lower levels of the natural anticoagulants protein C and free protein S.8
Recurrent bleeding is another concern. Intra-abdominal bleeding (ie, of pelvic or gastrointestinal origin) is the most concerning because it can be life-threatening. If there is large-volume blood flow through the lesions, high-output heart failure may occur due to the inability of the heart to generate sufficient cardiac output. Likewise, chronic lymphedema can lead to stasis dermatitis and recurrent episodes of cellulitis.9 Abnormal hematologic laboratory values are common—particularly thrombocytopenia, given the activation of intravascular coagulation. Our patient’s low platelet count at birth resolved before discharge home.
Pain is a common aspect of KTS; the prevalence ranges from 37% to 88%.9 And the associated limb-length discrepancies have long-term sequelae and may lead to scoliosis or impaired gait. Limb growth continues to adolescence, and limb-length differences greater than 2.5 cm at any age warrant a thorough evaluation.10
Treatment includes medical and surgical options. Compression stockings are recommended early on to prevent sequelae of venous stasis and lymphedema. Pulsed-dye laser therapy has also been successful.11 Sclerotherapy involves the injection of a chemical into the abnormal vein to cause thickening and obstruction. Treatment with oral propranolol may be promising; a case series of 2 patients with KTS showed reduced edema with use of the medication.12 However, in conjunction with supportive therapies, medical and surgical therapies aimed at reducing the burden of vascular malformations are successful at managing coagulopathy, pain, and infection.11
- Paller AS, Mancini AJ. Vascular disorders of infancy and childhood. In: Paller AS, Mancini AJ. Hurwitz Clinical Pediatric Dermatology: A Textbook of Skin Disorders of Childhood and Adolescence. 4th ed. Philadelphia, PA: Elsevier Saunders; 2011:chap 12.
- Paller AS, Mancini AJ. Skin disorders due to fungi. In: Paller AS, Mancini AJ. Hurwitz Clinical Pediatric Dermatology: A Textbook of Skin Disorders of Childhood and Adolescence. 4th ed. Philadelphia, PA: Elsevier Saunders; 2011:chap 17.
- Paller AS, Mancini AJ. Cutaneous tumors and tumor syndromes. In: Paller AS, Mancini AJ. Hurwitz Clinical Pediatric Dermatology: A Textbook of Skin Disorders of Childhood and Adolescence. 4th ed. Philadelphia, PA: Elsevier Saunders; 2011:chap 9.
- Berry SA, Peterson C, Mize W, et al. Klippel-Trenaunay syndrome. Am J Med Genet. 1998;79(4):319-326.
- Pereira C, Santo RE, Saldanha J. Klippel-Trenaunay syndrome: diagnosis in a neonate. BMJ Case Rep. 2017;2017. doi:10.1136/bcr-2017-221011.
- Luks VL, Kamitaki N, Vivero MP, et al. Lymphatic and other vascular malformative/overgrowth disorders are caused by somatic mutations in PIK3CA. J Pediatr. 2015;166(4):1048-1054.e1-5.
- Baskerville PA, Ackroyd JS, Lea Thomas M, et al. The Klippel-Trenaunay syndrome: clinical, radiological and haemodynamic features and management. Br J Surg. 1985;72(3):232-236.
- Oduber CEU, van Beers EJ, Bresser P, van der Horst CMAM, Meijers JCM, Gerdes VEA. Venous thromboembolism and prothrombotic parameters in Klippel-Trenaunay syndrome. J Med. 2013;71(5):246-252.
- Jacob AG, Driscoll DJ, Shaughnessy WJ, Stanson AW, Clay RP, Gloviczki P. Klippel-Trénaunay syndrome: spectrum and management. Mayo Clin Proc. 1998;73(1):28-36.
- Spencer SA, Sorger J. Orthopedic issues in vascular anomalies. Semin Pediatr Surg. 2014;23(4):227-232.
- Rahimi H, Hassannejad H, Moravvej H. Successful treatment of unilateral Klippel-Trenaunay syndrome with pulsed-dye laser in a 2-week old infant. J Lasers Med Sci. 2017;8(2):98-100.
- Wu JK, Hooper ED, Laifer-Narin SL, Simpson LL, Kandel J, Shawber CJ; Lymphatics Work Group at CUMC. Initial experience with propranolol treatment of lymphatic anomalies: a case series. Pediatrics. 2016;138(3):e20154545.


