Peer Reviewed
Purpura, Arthralgia, and Acute Kidney Injury in a Patient With Sepsis
Authors:
Colin F. Nolan, MD
Carrier Air Wing Seven, Naval Air Station Oceana, Virginia Beach, VirginiaWarren Ross, MD
Department of Radiology, Walter Reed National Military Medical Center, Bethesda, MarylandAnis Miladi, MD
Department of Dermatology, Naval Medical Center Portsmouth, Portsmouth, VirginiaCitation:
Nolan CF, Ross W, Miladi A. Purpura, arthralgia, and acute kidney injury in a patient with sepsis. Consultant. 2020;60(4):19-21, 24. doi:10.25270/con.2020.04.00004Disclaimer:
The views expressed in this article are those of the author(s) and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the United States Government.
A 51-year-old woman was hospitalized with sepsis secondary to tubo-ovarian abscess. The patient received intravenous ampicillin-sulbactam and underwent percutaneous drainage, which yielded 55 mL of purulent fluid. Intravenous ciprofloxacin was added to broaden antibiotic coverage.
Three days after the procedure, the patient exhibited persistent fevers with new-onset loose bowel movements and unilateral wrist pain. She also developed a purpuric, nonblanching, mildly pruritic rash affecting her extremities and, to a lesser extent, her trunk (Figure 1). There was no mucosal involvement, cutaneous ulceration, or focal necrosis of the dermis.
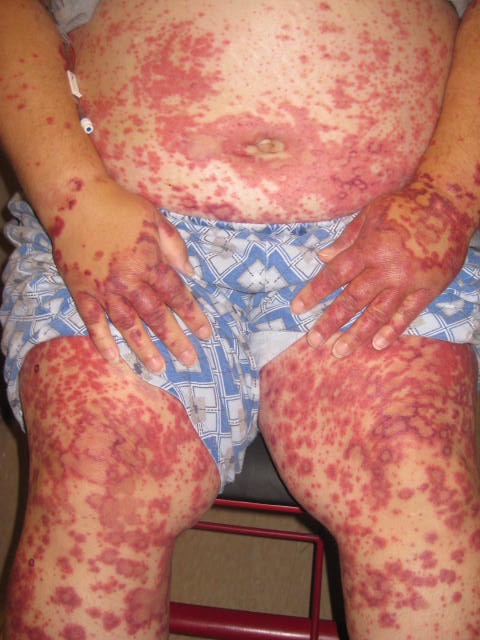
Figure 1. Palpable, purpuric patches and plaques affecting the trunk and the upper and lower extremities in a patient 3 days after administration of intravenous ciprofloxacin.Laboratory analysis revealed anemia without leukocytosis and an acute kidney injury. The serum creatinine level was 1.5 mg/dL. Urinalysis findings were negative for blood, although proteinuria, glycosuria, and hyaline casts were present. The erythrocyte sedimentation rate was mildly elevated at 45 mm/h. Study results for Clostridium difficile were negative.
The patient had a history of poorly controlled diabetes mellitus with bilateral peripheral neuropathy of the feet (a recent hemoglobin A1c concentration was 9.0%), hypertension, hypothyroidism, and hyperlipidemia. Her surgical history included laparoscopic cholecystectomy, laparoscopic appendectomy, and bilateral carpal tunnel release. She had a known allergy to sulfa-containing drugs. Her preadmission medication regimen included levothyroxine, insulin glargine, topiramate, montelukast, esomeprazole, metformin, pregabalin, cetirizine, duloxetine, and losartan.
What is your diagnosis?
A. Stevens-Johnson syndrome (SJS)
B. Drug reaction with eosinophilia and systemic symptoms (DRESS)
C. Eosinophilic granulomatosis with polyangiitis (Churg-Strauss syndrome)
D. Leukocytoclastic vasculitis (LCV)
E. Henoch-Schönlein purpura (HSP)Answer and discussion on next page.


