Atypical Celiac Disease: Could You Be Missing This Common Problem?
ABSTRACT: For every recognized case of celiac disease, 8 more remain undiagnosed. The reason for this disparity is contingent on the varying presentations of the disease. What was once considered solely a GI disorder, uniformly presenting with diarrhea and malabsorption, has evolved into a multisystem autoimmune disorder with myriad symptoms and signs. In addition, celiac disease is no longer a disorder limited to childhood and adolescence; it has even been diagnosed for the first time in elderly patients. Atypical celiac disease can be found in patients who present primarily with hypothyroidism, liver enzyme elevations, Addison disease, type 1 diabetes mellitus, and other disorders with major pathology outside the GI tract. Studies have demonstrated that an active casefinding strategy in primary care practice can increase the diagnostic yield for celiac disease.
_______________________________________________________________________________________________________________________________________________________
If you were to ask other primary care practitioners which subspecialty niche celiac disease should occupy, most would probably respond, "Gastroenterology, of course." Until recently, we would have agreed. However, celiac disease or, better yet, celiac syndrome needs to be "reframed" for contemporary clinicians.
For example, "atypical" celiac disease—which presents with few or even no GI symptoms or signs—is largely responsible for the increased prevalence of celiac disease today.1 Extraintestinal manifestations of celiac disease are rapidly becoming the rule rather than the exception. One example is thyroid disease, which occurs 4.4 times more frequently in patients with celiac disease than it does in control populations.2 In some of these persons, the thyroid disorder may be diagnosed and treated appropriately, but celiac disease may be either overlooked or only diagnosed later.
Celiac disease can be associated with a variety of autoimmune diseases including type 1 diabetes mellitus, autoimmune liver disease, Graves disease, and Addison disease.3 Thus, celiac disease has become one of our generation's great "masqueraders," similar to the role syphilis played in William Osler's day.

Figure 1 – Duodenal biopsy reveals severe villous atrophy–the classic pathology in celiac disease.
(Courtesy of CPT John Godino, MC, USA.)
Because of the myriad and frequently enigmatic presentations of celiac disease, the challenge of diagnosis falls squarely on the shoulders of primary care practitioners. Most cases are currently undiagnosed; however, one study demonstrated that an active case-finding strategy in primary care practices improves the detection rate.3
The study included 737 women and 239 men older than 40 years. Any participant with a family history of celiac disease or unexplained anemia or iron deficiency, recurrent abdominal pain or bloating, irritable bowel syndrome (IBS), chronic fatigue, unexplained liver enzyme abnormalities, or an autoimmune disease was offered serological screening for celiac disease. Those with a positive result were referred to a gastroenterologist for duodenal biopsy. The result of this approach was a 32- to 43-fold increase in the case finding of celiac disease.3
This study demonstrated that reframing celiac disease as a systemic autoimmune disorder is the key to earlier diagnosis. Here we will discuss how this approach can be used in primary care practice to augment case finding and appropriate consultation. We will present 3 case studies that illustrate atypical presentations of the disease, and we will also describe high-yield screening studies.
A CONTEMPORARY DIAGNOSTIC PARADIGM FOR CELIAC DISEASE

Figure 2 – This intensely pruritic rash on a man's leg is dermatitis herpetiformis, a cutaneous manifestation of celiac disease.
(Courtesy of Charles E. Crutchfield III, MD, and Eric J. Lewis, MD.)
The average delay in the diagnosis of celiac disease may be as long as 13 years.4 Moreover, for every recognized case, 8 more remain undiagnosed.4 Clearly, the current approach to celiac disease is in need of an update.
Broadening the definition. Celiac disease is no longer considered a disease limited to childhood or adolescence. It can present in any agegroup and has even been diagnosed for the first time in elderly patients (Case 1).
CLINICAL BACKGROUND
You evaluate a 76-year-old woman for the first time at a local nursing home. Her medical history includes iron deficiency anemia, osteopenia, and hypothyroidism. Her only surgery was a hysterectomy for uterine fibroids 30 years earlier. She takes iron supplements, a bisphosphonate, and thyroid hormone replacement therapy. She feels generally well, although she reports fatigue and decreased energy. Her weight has been stable, and she is active in her nursing home community. A review of systems is otherwise negative, and physical findings are unremarkable.
A previous workup for iron deficiency revealed no GI source of bleeding. Multiple fecal occult blood tests were negative, and the results of 2 colonoscopies and upper endoscopies were normal. There are no other apparent sources of blood loss; she had a hysterectomy years earlier, and her urine has never contained gross or microscopic blood.
Despite the patient's adherence to the iron replacement regimen, laboratory values continue to be abnormal (hemoglobin, 10.4 g/dL; red blood cell distribution width, 20%; mean corpuscular volume, 70 μm3; and ferritin, 20 ng/mL). You suspect that iron deficiency is limiting her quality of life. How would you proceed?
CASE DISCUSSION
The duodenum is the anatomical site where iron is absorbed from the GI tract. This patient's primary care physician ordered initial serological screening tests for IgA anti–tissue-transglutaminase and IgA anti-endomysial antibodies, which were positive. An endoscopy with 6 separate duodenal biopsies confirmed celiac disease.
A gluten-free diet was prescribed. The patient's ferritin level increased, and the anemia and microcytosis resolved. She also reported increased energy. It was suspected that celiac disease might have contributed to her osteopenia and hypothyroidism as well.
TAKE-HOME MESSAGE
The prevalence of celiac disease is increasing among elderly patients.44 In fact, 25% of patients in one study received a diagnosis of celiac disease in their seventh decade.45
Consider atypical celiac disease in elderly patients with unexplained anemia, osteopenia, or endocrine disease. Such patients may lack classic GI malabsorptive symptoms and signs.
Elderly persons with celiac disease may have low folic acid and vitamin B12 levels or a calcium deficiency from occult intestinal malabsorptive abnormalities.44 Like their younger counterparts, elderly persons with celiac disease may present with dermatitis herpetiformis (Figure 2). In some older adults, biopsy-proven collagenous colitis may suggest celiac disease.46
In a cohort of patients with celiac disease who also had dementia, a gluten-free diet improved cognitive deficiency.47 Keep in mind that the gluten filler in medication capsules may cause relapse in some treated patients.44
Since Samuel Gee's 1887 description of diarrhea as the only classic symptom of celiac disease,5 diarrhea has been accepted as a "universal symptom." If a patient did not have diarrhea, the physician did not think of celiac disease.
This era's challenge is to realize that celiac disease frequently presents without diarrhea. Today, the disease is a broader syndrome without a single or universal symptom; instead, it manifests itself as a systemic illness in organs outside the GI tract. This phenomenon is evidenced by the robust attention paid to extraintestinal celiac disease in recent literature.6-8 Before 1980, about 71% of patients in whom celiac disease was diagnosed had diarrhea. More recently, the incidence of diarrhea as a manifestation of the disease has dropped to 37%.9
Updated classification. Celiac disease can be classified as typical (classic or symptomatic with intestinal symptoms and signs), atypical (with extraintestinal or atypical symptoms and signs), or silent/latent (completely asymptomatic and, as a result, undiagnosed). This classification represents an updated diagnostic paradigm for the disease.
Symptoms of typical celiac disease include abdominal pain, often crampy in nature and associated with distention; dyspepsia and other symptoms of gastroesophageal reflux disease; and altered bowel habits accompanied by diarrhea. Patients with typical celiac disease frequently experience weight loss, anemia (from iron or vitamin B12 deficiency), and fatigue, and they can have osteopenia.7
A newer observation regarding typical celiac disease is that it can mimic IBS. A study showed that of 105 patients presumed to have IBS, 11.4% actually had celiac disease. After appropriate diagnostic studies, these patients were treated for celiac disease, rather than for IBS, and their symptoms abated.10 Another British study evaluated about 300 patients with IBS and found 22% of them had specific antibodies that suggested celiac disease. Later, 4.7% of these patients were unequivocally confirmed to have celiac disease by intestinal biopsies.11
Atypical celiac disease is characterized by the absence of "typical" GI manifestations. Patients present with signs previously considered secondary or unusual accompaniments of celiac disease, such as iron deficiency or elevated liver enzyme levels (Case 1 and Case 2).
CLINICAL BACKGROUND
A 16-year-old girl comes to your office because of persistent, mild liver enzyme elevations. On 2 occasions during the past 6 months, she has had aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels of 80 U/L and 92 U/L and 94 U/L and 90 U/L, respectively. She is otherwise healthy except for seasonal allergies. She takes no medications (either over-the- counter or prescription), does not use any alternative therapies, and does not drink alcohol or use illicit drugs. She does not recall ever having jaundice or hepatitis. There is no significant past medical or family history. She is an athlete and is not obese (body mass index, 21). Physical findings are normal for her age.
Additional laboratory testing reveals the following values: normal complete blood cell count, continued similar elevations of AST and ALT, and normal metabolic parameters (including glucose and triglycerides). Other liver-related studies—albumin, total protein, bilirubin, and alkaline phosphatase levels—are normal. An ultrasound examination demonstrates a normal liver without suggestion of steatosis.
A second set of laboratory studies is ordered to further investigate the persistent elevations in AST and ALT. Viral causes are excluded by normal results for hepatitis A, B, and C panels; as well as measurement of Epstein-Barr virus and cytomegalovirus antibody levels. The ceruloplasmin level is also normal. An autoimmune liver disease panel (antinuclear antibody and anti–kidney/liver microsome, anti–liver cytosol, and anti–smooth muscle antibodies) is negative. Evaluation for α1-antitrypsin deficiency is also negative. Would you test this patient for celiac disease?
CASE DISCUSSION
She should definitely undergo serological testing (anti–tissue-transglutaminase and anti-endomysial antibodies) for latent or atypical celiac disease. The most common hepatic presentation of celiac disease is an isolated elevation of AST and ALT (typically less than 5 times the upper limit of normal values).48 In fact, after workup for unexplained liver enzyme elevations, about 1 in 10 persons are found to have celiac disease.49
TAKE-HOME MESSAGE
Celiac disease is also associated with more severe liver disease, including an 8-fold increase in the risk of cirrhosis.49 The clinical connection between celiac disease and primary biliary cirrhosis may be related to a common immunological mechanism.49 Other studies have demonstrated a relationship between celiac disease and autoimmune hepatitis and primary sclerosing cholangitis. Consensus guidelines strongly recommend that serological tests for celiac disease be included in the comprehensive screening of persons with liver enzyme elevations.50
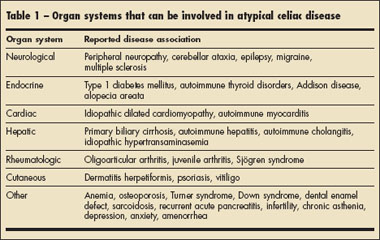
It is the autoimmune characteristics of celiac disease that make it a multisystem disorder. The antibodies associated with celiac disease target multiple organs outside the GI tract; Table 1 highlights the various organ systems that can be involved.6,7 Case 3 illustrates a subtle presentation of atypical celiac disease in primary care practice.
Silent celiac disease occurs in persons who are asymptomatic but who have a positive serological test and villous atrophy on biopsy—the classic pathology of celiac disease (Figure 1). Testing is performed because the patient has relatives with celiac disease.
CLINICAL BACKGROUND
A man in his 30s presented to the emergency department (ED) with hypotension (95/60 mm Hg). He had a 10-year history of type 1 diabetes mellitus, and his most recent hemoglobin A1c (HbA1c) level was 7.4%. Infection and sepsis were initially suspected, but the serum sodium and potassium levels were 110 mEq/L and 7.0 mEq/L, respectively. The ED physician noted that the patient was "pigmented."
The combination of hypotension and hyperpigmentation in a patient with type 1 diabetes mellitus accompanied by characteristic electrolyte abnormalities suggested an underlying autoimmune syndrome manifested by Addison disease. The patient was treated vigorously with normal saline as well as parenteral and later oral corticosteroids, and his condition improved. A random serum cortisol concentration was 1 μg/dL and increased to 3 μg/dL only after stimulation with parenteral adrenocorticotropic hormone (ACTH). The patient continued to do well with insulin and oral prednisone.
Many years later, he comes to you with well-controlled type 1 diabetes mellitus (HbA1c, 6.9%) and Addison disease that has not caused further problems. He is found to have iron deficiency (hemoglobin level, 11.0 g/dL; red blood cell distribution width, 18%; mean corpuscular volume, 69 μm3; and ferritin level, 15 ng/mL). The patient has no GI complaints, and 3 fecal occult blood tests are negative. His thyroid-stimulating hormone level is high, consistent with hypothyroidism. Tests for antithyroid antibodies are positive. How would you proceed?
CASE DISCUSSION
Celiac disease is an autoimmune disorder. Affected patients produce highly disease-specific IgA and IgG autoantibodies to tissue transglutaminase. Their own lymphocytes can be cytotoxic to intestinal and many other cells in the body.51
The prevalence of celiac disease is higher among persons with type 1 diabetes mellitus (5.7%), also an autoimmune disease, as well as among relatives of those with type 1 diabetes (1.9%), compared with age-matched controls.52 More recent data have demonstrated that persons with type 1 diabetes mellitus and those with celiac disease share a common genetic background.53 In fact, the 2 diseases share 7 genetic regions. This patient's antibody screening tests were positive for celiac disease, and he was referred to a gastroenterologist.
TAKE-HOME MESSAGE
Consider celiac disease as an underlying pathology in patients with autoimmune endocrine diseases, such as type 1 diabetes mellitus, hypothyroidism, and Addison disease, whether or not diarrhea is present.
Latent celiac disease is defined by a normal duodenal biopsy result in a person with a positive serology and family history who is consuming a regular (gluten-containing) diet. Villous atrophy develops in such persons later in life. Although many persons with latent celiac disease are asymptomatic, some have symptoms and therefore may present in the primary care setting.12
PATHOGENESIS OF CELIAC DISEASE
Role of heredity. HLA complex class II genes are involved in the pathogenesis of celiac disease.13 Louka and Sollid14 have implicated the inheritance of HLA-DQA1*05- DQB1*02 (DQ2) and DQA1*03- DQB1*0302 (DQ8) as a predisposition to the development of celiac disease. International twin studies have also demonstrated that there is a 70% to 85% concordance rate of celiac disease in monozygotic twins and a 10% to 20% rate in dizygotic twins.15,16
HLA genes contribute approximately 53% to the total prevalence of celiac disease.17 Thus, the disease does not develop in all persons who have "at-risk" HLA genes; the role of non-HLA genes is also vital to its pathogenesis. Currently, the non- HLA genes that are potentially associated with celiac disease include COELIAC2 (5q31-33), COELIAC3 (2q33), and COELIAC4 (19p13.1).18-20 The 11q locus also has been documented to confer some degree of susceptibility to the eventual development of the disease.21
Environmental triggers. The role of environmental factors is as important as a genetic predisposition to celiac disease. The putative role of a gluten-rich diet is well known. Gluten is the storage portion of wheat protein. It has 2 major components: gliadins and glutenins. Other proteins associated with celiac disease may be found in barley (hordeins) and rye (scalins). Oats are not typically implicated as a trigger for celiac disease. Of all the potential triggers for celiac disease, glutenins have the strongest association.22-24
Kagnoff25 detailed the following series of events as a plausible environmental pathogenesis for celiac disease. When a genetically predisposed person (who has HLA and/or other candidate genes) ingests a gluten-rich diet, the proline content of gluten resists complete digestion. As a result, it accumulates within undigested gluten peptides. These undigested peptides cross the intestinal epithelial barrier and reach the lamina propria. There they come in contact with tissue transglutaminase (t-TGase) and antigen-presenting cells (APCs).
APCs express HLA-DQ2 or HLADQ8 heterodimers, which have the capacity to bind to t-TGase. Once this binding occurs, APCs present the bound combination to CD4+ T cells specific for HLA-DQ2 and HLA-DQ8. These T cells release inflammatory mediators and cause villous injury.25
Possible mechanism for extraintestinal manifestations. Tissue-TGase assists in cell-matrix interactions and thus plays a vital role in building extracellular matrix. This enzyme is present in nearly every organ throughout the body; it is not limited to the GI tract.26,27
Tissue-TGase gluten residues may produce generalized antigenic epitopes through cross-linking undigested gliadin peptides.22 These peptides then cross intestinal barriers, reaching the lamina propria. There the complexes undergo endocytosis, further processed by specific B cells. These B cells present the complexes to gliadin-specific T cells, triggering immune reactions against t-TGase.28,29 The autoantibodies are known as anti–tissue-transglutaminase (anti–t-TGase) antibodies. Since t-TGase is ubiquitous, these antibodies have the potential to injure multiple organs through inflammatory reactions.
SCREENING FOR CELIAC DISEASE IN PRIMARY CARE PRACTICE
Older diagnostic criteria. The European Society of Pediatrics, Gastroenterology, and Nutrition (ESPGAN) published diagnostic criteria for celiac disease in 197030,31 and modified them in 1990 (Table 2).31,32 The criteria initially focused on celiac disease as a GI disorder and were also intended primarily for children.

A new diagnostic approach. Serological tests have become essential to the diagnosis of celiac disease. The arrays available in the primary care setting have included IgA antigliadin antibody, IgA antireticulin, IgA antiendomysial antibody (EMA) and, more recently, anti–t-TGase antibody. 33-38 IgA antigliadin antibody was the earliest screening choice. Screening for IgA anti–t-TGase antibodies, coupled with EMA, confers sensitivities of 98% to 100%, specificities of 98% to 100%, a positive predictive value of 90%, and a negative predictive value of 95% for celiac disease.4
The current American Gastroenterological Association guidelines support the use of IgA anti–t-TGase antibody and not EMA as a screening test for celiac disease.39 EMA is a confirmative test and should not be used for screening because of its lower sensitivity. Antireticulin is not recommended as a screening test.
Who should be screened? In addition to patients with typical GI manifestations, anyone who has a potential atypical presentation of celiac disease can benefit, such as patients with unexplained endocrine autoimmunity or IBS. Also consider screening for latent disease in persons who have a positive family history. Refer patients with positive antibody screening results for endoscopy and duodenal biopsy.
An experienced laboratory is required for diagnostic accuracy. Certain variables may contribute to the predictive power of the antibody screening tests. For example, tests that rely on IgA for diagnosis might be inappropriately negative (false-negative) in patients with both celiac disease and IgA deficiency. In fact, celiac disease is more common in persons with IgA deficiency than in the general population.40 IgG serological tests (IgG endomysial antibodies, IgG anti–t-TGase, IgG antigliadin antibody) are available for use in this specific population.
Some patients with suspected celiac disease may have negative serology results because of liver failure, Sjögren syndrome, or immunosuppressive therapy, for example. If suspicion is high, endoscopy can be performed in such patients.
CONSEQUENCES OF UNDIAGNOSED CELIAC DISEASE
What are the risks to patients if celiac disease is latent or atypical and remains undiagnosed? A recent study showed that undiagnosed celiac disease is associated with a 4-fold increase in all-cause mortality.41 In addition, patients with untreated celiac disease have a lower response rate to vaccinations such as hepatitis B.
Another study revealed that patients with celiac disease have a 5-fold higher risk of non-Hodgkin lymphoma.42 In this study, family members of patients with celiac disease, who themselves did not have the disease, also had a higher risk of lymphoma. Moreover, celiac disease is associated with an almost 4 times greater risk of tuberculosis.43
Celiac disease may be silent or atypical, but it is still a serious disorder. It is in the best interests of our patients and their families to suspect celiac disease in all its recently discovered disguises.
1. Guthrie EW, Gettis MA. Celiac disease: more common than once thought. US Pharm. 2008;33: 24–29. http://www.uspharmacist.com/content/t/gastroenterology/c/11776. Accessed January 15, 2010.
2. Elfström P, Montgomery SM, Kämpe O, et al. Risk of thyroid disease in individuals with celiac disease. J Clin Endocrinol Metab. 2008;93:3915–3921.
3. Catassi C, Kryszak D, Louis–Jacques O, et al. Detection of celiac disease in primary care: a multicenter case–finding study in North America. Am J Gastroenterol. 2007;102:1454–1460.
4. Leeds JS, Hopper AD, Sanders DS. Coeliac disease. Am J Gastroenterol. 2008;88:157–170.
5. Gee S. On the coeliac affection. St Bartholomew Hospital Rep. 1888;24:17–20.
6. Lee SK, Green PH. Celiac sprue (the great modern–day imposter). Curr Opin Rheumatol. 2006;18:101–107.
7. Rodrigo L. Celiac disease. World J Gastroenterol. 2006;12:6585–6593.
8. Hernandez L, Green PH. Extraintestinal manifestations of celiac disease. Curr Gastroenterol Rep. 2006;8:383–389.
9. Craig D, Robins G, Howdle PD. Advances in celiac disease. Curr Opin Gastroenterol. 2007;23:142–148.
10. Shahbazkhani B, Forootan M, Merat S, et al. Coeliac disease presenting with symptoms of irritable bowel syndrome. Aliment Pharmacol Ther. 2003;18:231–235.
11. Sanders DS, Carter MJ, Hurlstone DP, et al. Association of adult coeliac disease with irritable bowel syndrome: a case control study in patients fulfilling ROME II criteria referred to secondary care. Lancet. 2001;358:1504–1508.
12. Ludvigsson JF, Brandt L, Montgomery SM. Symptoms and signs in individuals with serology positive for celiac disease but normal mucosa. BMC Gastroenterol. 2009;9:57.
13. Sollid LM, Thorsby E. HLA susceptibility genes in celiac disease: genetic mapping and role in pathogenesis [published correction appears in Gastroenterology. 1994;106:1133]. Gastroenterology. 1993;105:910–922.
14. Louka AS, Sollid LM. HLA in coeliac disease: unravelling the complex genetics of a complex disorder. Tissue Antigens. 2003;61:105–117.
15. Greco L, Romino R, Coto I, et al. The first large population based twin study of coeliac disease. Gut. 2002;50:624–628.
16. Nisticò L, Fagnani C, Coto I, et al. Concordance, disease progression, and heritability of coeliac disease in Italian twins. Gut. 2006;55:803–808.
17. Sollid LM, Lie BA. Celiac disease genetics: current concepts and practical applications. Clin Gastroenterol Hepatol. 2005;3:843–851.
18. Greco L, Corazza G, Babron MC, et al. Genome search in celiac disease. Am J Hum Genet. 1998;62:669–675.
19. Holopainen P, Naluai AT, Moodie S, et al; Members of the European Genetics Cluster on Coeliac Disease. Candidate gene region 2q33 in European families with coeliac disease. Tissue Antigens. 2004;63:212–222.
20. Monsuur AJ, de Bakker PI, Alizadeh BZ, et al. Myosin IXB variant increases the risk of celiac disease and points toward a primary intestinal barrier defect. Nat Genet. 2005;37:1341–1344.
21. Naluai AT, Nilsson S, Gudjónsdóttir AH, et al. Genome–wide linkage analysis of Scandinavian affected sib–pairs supports presence of susceptibility loci for celiac disease on chromosomes 5 and 11. Eur J Hum Genet. 2001;9:938–944.
22. van de Wal Y, Kooy YM, van Veelen P, et al. Glutenin is involved in the gluten–driven mucosal T cell response. Eur J Immunol. 1999;29:3133–3139.
23. Molberg Ø;, Solheim Flaete N, Jensen T, et al. Intestinal T–cell responses to high–molecular–weight glutenins in celiac disease. Gastroenterology. 2003; 125:337–344.
24. Dewar DH, Amato M, Ellis HJ, et al. The toxicity of high molecular weight glutenin subunits of wheat to patients with coeliac disease. Eur J Gastroenterol Hepatol. 2006;18:483–491.
25. Kagnoff MF. Celiac disease: pathogenesis of a model immunogenetic disease. J Clin Invest. 2007;117:41–49.
26. Aeschlimann D, Paulsson M. Transglutaminases: protein cross–linking enzymes in tissues and body fluids. Thromb Haemost. 1994;71:402–415.
27. Greenberg CS, Birckbichler PJ, Rice RH. Transglutaminases: multifunctional cross–linking enzymes that stabilize tissues. FASEB J. 1991;5:3071–3077.
28. Molberg O, McAdam SN, Sollid LM. Role of tissue transglutaminase in celiac disease. J Pediatr Gastroenterol Nutr. 2000;30:232–240.
29. Sollid LM, Molberg O, McAdam S, Lundin KE. Autoantibodies in coeliac disease: tissue transglutaminase—guilt by association? Gut. 1997;41:851–852.
30. Meuwisse G. Diagnostic criteria in coeliac disease. Acta Paediatr Scand. 1970;59:461–463.
31. Stenhammar L, Högberg L, Danielsson L, et al. How do Swedish paediatric clinics diagnose coeliac disease? Results of a nationwide questionnaire study. Acta Paediatr. 2006;95:1495–1497.
32. European Society of Paediatric Gastroenterology and Nutrition. Revised criteria for diagnosis of coeliac disease. Report of Working Group of European Society of Paediatric Gastroenterology and Nutrition. Arch Dis Child. 1990;65:909–911.
33. Savilahti E, Viander M, Perkkiö M, et al. IgA antigliadin antibodies: a marker of mucosal damage in childhood coeliac disease. Lancet. 1983;1:320–322.
34. Unsworth DJ, Walker–Smith JA, Holborow EJ. Gliadin and reticulin antibodies in childhood coeliac disease. Lancet. 1983;1:874–875.
35. Mäki M, Häliström O, Vesikari T, Visakorpi JK. Evaluation of a serum IgA–class reticulin antibody test for the detection of childhood celiac disease. J Pediatr. 1984;105:901–905.
36. Bürgin–Wolff A, Gaze H, Lentze M, Nusslé D. Gliadin and endomysium antibody determinations in childhood celiac disease. In: Kumar P, Walker–Smith JA, eds. Proceedings of International Coeliac Symposium. Leeds, UK: Leeds University Press; 1990.
37. Chorzelski TP, Beutner EH, Sulej J, et al. IgA anti–endomysium antibody. A new immunologic marker of dermatitis herpetiformis and coeliac disease. Br J Dermatol. 1984;111:395–402.
38. Sblattero D, Berti I, Trevisiol C, et al. Human recombinant tissue transglutaminase ELISA: an innovative diagnostic assay for celiac disease. Am J Gastroenterol. 2000;95:1253–1257.
39. AGA (American Gastroenterological Association) Institute. AGA Institute medical position statement on the diagnosis and management of celiac disease. Gastroenterology. 2006;131:1977–1980.
40. Webster AD, Slavin G, Shiner M, et al. Coeliac disease with severe hypogammaglobulinaemia. Gut. 1981;22:153–157.
41. Rubio–Tapia A, Kyle RA, Kaplan EL, et al. Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology. 2009;137:88–93.
42. Gao Y, Kristinsson SY, Goldin LR, et al. Increased risk for non–Hodgkin lymphoma in individuals with celiac disease and a potential familial association. Gastroenterology. 2009;136:91–98.
43. Ludvigsson JF, Wahlstrom J, Grunewald J, et al. Coeliac disease and risk of tuberculosis: a population based cohort study. Thorax. 2007;62:23–28.
44. Freeman HJ. Adult celiac disease in the elderly. World J Gastroenterol. 2008;14:6911–6914.
45. O'Morain C, Segal AW, Levi AJ. Elemental diets in treatment of acute Crohn's disease. Br Med J. 1980:281:1173–1175.
46. Freeman HJ. Collagenous colitis as the presenting feature of biopsy–defined celiac disease. J Clin Gastroenterol. 2004;38:664–668.
47. Patel D, Kalkat P, Baisch D, Zipser R. Celiac disease in the elderly. Gerontology. 2005;51:213–214.
48. Díaz R, Kelly JA, Ruchelli E, et al. A 10–year–old girl with mild elevation of liver transaminases. Medscape J Med. 2008;10:28.
49. Rubio–Tapia A, Murray JA. The liver in celiac disease. Hepatology. 2007;46:1650–1658.
50. Morisco F, Pagliaro L, Caporaso N, et al; University of Naples Federico II, Italy. Consensus recommendations for managing asymptomatic persistent non–virus non–alcohol related elevation of aminotransferase levels: suggestions for diagnostic procedures and monitoring. Dig Liver Dis. 2008;40:585–598.
51. Fasano A. Systemic autoimmune disorders in celiac disease. Curr Opin Gastroenterol. 2006;22: 674–679.
52. Ferguson J, Brownlee D, Howie K, Campbell IW. Coeliac disease: relationship to endocrine autoimmunity. Br J Diabetes Vasc Dis. 2006;6:287–288.
53. Smyth DJ, Plagnol V, Walker NM, et al. Shared and distinct genetic variants in type 1 diabetes and celiac disease. N Engl J Med. 2008;359:2767–2777.


